Print Entire Issue
ICHP Info

Illinois Council of Health-System Pharmacists
4055 North Perryville Road
Loves Park, IL 61111-8653
Phone: (815) 227-9292
Fax: (815) 227-9294
ichpnet.org
KeePosted
Official News journal of the Illinois Council of Health-System Pharmacists
EDITOR
Jennifer Phillips
ASSISTANT EDITOR
Milena Murray
MANAGING EDITOR
Trish Wegner
ASSISTANT MANAGING EDITOR
Maggie DiMarco Allen
DESIGN EDITOR
Melissa Dyrdahl
ICHP Staff
EXECUTIVE VICE PRESIDENT
Chris Crank
VICE PRESIDENT - PROFESSIONAL SERVICES
Trish Wegner
DIRECTOR OF OPERATIONS
Maggie Allen
INFORMATION SPECIALIST
Heidi Sunday
CUSTOMER SERVICE AND
PHARMACY TECH TOPICS™ SPECIALIST
Jo Ann Haley
ACCOUNTANT
Kim Anderson
COMMUNICATIONS MANAGER
Melissa Dyrdahl
LEGISLATIVE CONSULTANT
Liz Brown Reeves
ICHP's Mission Statement
Advancing Excellence in
Pharmacy
ICHP's Vision Statement
ICHP dedicates itself to
achieving a vision of pharmacy practice where:
·
Pharmacists are
universally recognized as health care professionals and essential providers of
health care services.
·
Pharmacists use their
medication expertise and leadership skills to optimize the medication use
process and patient outcomes.
·
Pharmacy technicians are
trained and PTCB certified to manage the medication distribution process.
ICHP's Goal Statements
·
Raising awareness of the
critical role pharmacists fulfill in optimizing medication therapy and ensuring
medication safety in team-based, patient-centered care.
·
Providing high quality
educational services through innovative continuing pharmacy education and
training programs, and sharing evidence-based best practices.
·
Developing and nurturing
leaders through mentorship, skill development programs, and leadership
opportunities.
·
Working with national
and state legislators and policymakers to create or revise legislation and
regulation critical to pharmacy practice and quality patient care.
·
Urging pharmacy
technician employers to require successful completion of an accredited pharmacy
technician training program and PTCB certification of all pharmacy technicians.
Approved by the ICHP
Board of Directors May 30, 2018.
KeePosted Vision
As an integral publication of the Illinois Council of Health-System
Pharmacists, the KeePosted newsjournal will reflect its
mission and goals. In conjunction with those goals, KeePosted will
provide timely information that meets the changing professional and personal
needs of Illinois pharmacists and technicians, and maintain high publication
standards.
KeePosted is an official publication of, and is copyrighted by, the
Illinois Council of Health-System Pharmacists (ICHP). KeePosted is
published 4 times a year. ICHP members received KeePosted as
a member benefit. All articles published herein represent the opinions of the
authors and do not reflect the policy of the ICHP or the authors’ institutions
unless specified. Advertising inquiries can be directed to ICHP office at the
address listed above. Image disclaimer: The image used in the Pharmacy Tech
Topics™ advertisement is the property of © 2017 Thinkstock, a division of Getty
Images. Some images are property of © 2021 Adobe Stock.
Copyright © 2021, Illinois Council of Health-System Pharmacists. All rights
reserved.
ICHP Info
Columns
President's Message
Crank's Corner
Board of Pharmacy Update
Government Affairs
Professional Affairs
Professional Affairs
Professional Affairs
Professional Affairs - Best Practice Award Winner!
New Practitioners Network
Educational Affairs
ICHP Leadership Spotlight
ICHP Leadership Spotlight
ICHPeople
College Connection
University of Illinois at Chicago, College of Pharmacy
Midwestern University College of Pharmacy, Downers Grove
Southern Illinois University Edwardsville School of Pharmacy
Features
2021 ICHP Annual Meeting Recap
More
Upcoming Events
Welcome New Members!
ICHP Pharmacy Action Fund Contributors (PAC)
Board of Directors
Columns
 President's Message
President's Message
Everybody In
by Kathryn Schultz, PharmD, BCPS, BCOP; Malignant Hematology Medical Science Liaison; Genentech; President of ICHP
First, I would like to thank Jen Arnoldi for her leadership and guidance over the past year. It has been an exceptionally challenging year to serve as president for the organization, but Jen has handled it with poise, professionalism and by always putting members first while coming up with ways to continue to build bridges even though we are not physically together as we used to be.
Second, I would like to thank the many who have impacted my career in so many ways. Starting with my cousin, Jim Dorociak, who first introduced the concept of pharmacy as a career path to a fresh college graduate working in a molecular biology lab who was wondering what in the world to do with a bachelor’s degree in science that would let her speak with people instead of talking to cell cultures and pipettes. To my fellow pharmacy school students and professors from Midwestern University College of Pharmacy – where I learned the importance of didactic and experiential education as well as the importance of professional involvement – and where I first joined ICHP. I would also like to recognize and thank my co-residents, friends and colleagues from Rush University Medical Center and the University of Michigan. I wish I could be with you all in person this year and thank each and every one of you! There are just too many people to name and the impact of those experiences cannot even be put into words to describe how much they have shaped who I am as a clinician, a researcher, an advocate and a person. And finally, my ICHP network. ICHP has become a constant throughout my time in pharmacy school, residency and as a pharmacist. It is the backbone of pharmacy professionals banding together to advance the practice of pharmacy for ourselves and for our patients.
I think that’s a key take home point from this past year or year-plus, really. We can’t do this alone, there are many ways we interact and impact each other even when we are not working or meeting side by side, but we have built the bridges needed to work together. We have this common network to reach out to, collaborate with, and lean on both professionally and personally. Thinking about the integral pieces of the pharmacy, we have technicians - the gears that keep things churning in many pharmacy practices day after day. We have students - eager to learn as well as keeping preceptors on their toes! We have residents - striving for additional on-the-job education after pharmacy school as well as learning how to practice in a variety of new environments. We have pharmacists in many different areas of practice and settings, all coming together to teach each other (as we saw over these few days of the annual meeting) and pushing the boundaries of new and innovative patient care. Within these groups of collaborators, we have team captains and mentors - both formal and informal - providing history, guidance, experience and encouragement to continue to elevate the profession of pharmacy.
The thing is, we’re all in this together. Every single one of us. Whether it be compounding IVs, developing clinical pathways, providing education, overseeing clinical trials, planning and organizing conversion of units to care for surging needs or administration of vaccines! Everybody has an important role to fulfill. Everybody contributes to the greater good of the whole group. Everybody is in!
For those of you who know me, you are not surprised that my theme is a quote from Joe Maddon, who was the manager for the Chicago Cubs during their triumph in 2016 winning the World Series for the first time in 108 years! Joe Maddon believed in his team members, each and every one of them whether they were winning a championship or playing day to day games. Everybody In! He believed in the message to put the group ahead of the individuals.
That Cubs fans’ eternal optimism helps me to find the silver lining in anything. While it’s been a bit more challenging in this past year, we have shown what pharmacy can do as a profession when we band together. While sports usually make great analogies, I do think an inspirational non-sports story can provide an even better example to demonstrate the great strides that can be made in medicine when everybody is in.
There is a local story that depicts this essence of banding together people of different backgrounds and strengths with a common goal. It involves the University of Illinois. Their goal was to develop an efficient and effective screening test and tracing protocol to allow for the safe opening of University of Illinois schools. Not only did they develop an economical and expedient new screening test in record time, but they were able to test it and develop a protocol that will expand access to this test to school systems throughout the state of Illinois. They had chemists and molecular biologists working with research laboratories at the veterinary school, students and faculty providing saliva test samples and manufacturers collaborating with the suppliers for PCR testing reagents. EVERYBODY was in. Earlier this year it was reported that they had processed over 1.5 million tests, had received emergency use authorization, and had been able to partner with seven high-capacity labs across the state to help process the tests on a larger scale.1,2,3
Over the past year, we’ve also seen large phase 3 trials for vaccines start, finish, provide data for FDA review, and receive full approval for prevention of infection, hospitalization, and death. That timeline is unheard of. Additionally, many others have pushed the envelope by looking to repurpose available drugs and new molecules for the treatment of SARS-CoV2. Pharmacists, technicians, students, and residents have shown what can be done on the front lines to provide vaccines to the masses, while simultaneously caring for those who are ill and hospitalized for various conditions. In the state of Illinois, 6.64 million people are now vaccinated. This is what can be accomplished when we put the greater good of the group ahead of individuals.
I have another example of what can be accomplished when Everybody is in - this Annual Conference! The staff at ICHP, along with the Annual Meeting planning committee, have exemplified the theme for the next year by converting this conference to a successful virtual experience in an incredibly short amount of time. This couldn’t have been done unless Everybody (was) In! I want to give a special thank you to the ICHP staff: Maggie DiMarco Allen, Trish Wegner, Heidi Sunday, Melissa Dyrdahl, Kim Anderson, Jo Haley, Matthew Smith and Chris Crank along with the Annual Meeting Planning committee: Matt Allsbrook, Abby Kahaleh, Daniel Majerczyk, Becky Ohrmund, Marianne Pop, Natalie Tucker, Janki Vyas and the chair, David Martin. I seriously couldn’t have planted a better illustration to kick off next year’s theme if I tried!
So, what will we do next? What can we do next? What do we want to set our focus on to continue to show the world what the profession of pharmacy can achieve and continue to advocate for patient care? There really are no limits when Everybody is In.
References:
- How one university built a COVID-19 screening system https://cen.acs.org/biological-chemistry/infectious-disease/one-university-built-COVID-19/98/i42 [accessed 14 September 2021].
- Saliva-Based Coronavirus Test Developed by U of I Puts Illinois on ‘Cutting Edge,' Officials Say. https://www.nbcchicago.com/news/coronavirus/saliva-based-coronavirus-test-developed-by-u-of-i-puts-illinois-on-cutting-edge-officials-say/2325098/ [accessed 13 September 2021].
- University of Illinois saliva-based COVID 19 test gets FDA authorization https://abc7chicago.com/amp/covid-test-ilinois-testing-university-of-illinois/10380448/ [accessed 15 September 2021].
 Crank's Corner
Crank's Corner
Dealing With Disappointment
by Christopher W. Crank, PharmD, MS, BCPS; ICHP Executive Vice President
There has been a lot of opportunity to experience disappointment in 2020 and 2021. The disappointment has occurred in our professional and personal lives. Events have been cancelled or held virtually. We have had vacation plans postponed or cancelled in addition to limited opportunities to participate in family gatherings. All these reasons to experience disappointment during the pandemic are in addition to the usual disappointments that are a part of life.
I know I have been disappointed that I have still not been able to participate in a live educational meeting as ICHP’s Executive Vice President. I miss the interaction that you get with a live event. I have also been disappointed by the fact that I have not been able to take a road trip to visit health-systems across Illinois to network and promote ICHP. Based on my own disappointment, I decided to review what experts recommend. I have summarized my findings in a few key points.
Accept that it is OK to be disappointed.
It is normal to be sad or angry when you are disappointed. The goal should not be to never get upset. The goal should be to learn to manage and work through your disappointment. Acknowledge your frustrations by expressing them through writing or a conversation with a trusted friend. Expressing your feelings in a safe setting may help you overcome the feelings.
Keep things in perspective.
Try not to allow your disappointment to become your reality. Avoid negative thinking like “I guess we are never going to get to go on vacation” or “this is never going to end.” Keep your level of disappointment in line with how significant the issue is. When I am upset, I like to ask myself whether the problem will impact me in five years. If my answer is no, I try to reset my perspective and adjust my reaction to the situation accordingly.
Don’t dwell on your disappointment.
Wallowing in your disappointment won’t get you anywhere. Don’t beat yourself up or focus solely on the issue. Look for ways to move past it. Think of what you learned from the situation. Give yourself a pass on feeling upset but find ways to move on, such as by focusing on something else, such as a project or activity.
Set reasonable expectations next time.
It is important to ask if your expectations were reasonable to begin with. If you booked a vacation during the pandemic, you probably should have gone into it realizing that your plans might need to be altered based on changing situations. Multiple experts mentioned that higher hopes equal a bigger letdown when things don’t go as planned. I am not suggesting that you set low expectations so that you are always pleasantly surprised by better results; just keep your expectations reasonable.
Anticipate disappointment so you can manage it better.
If you can anticipate disappointment, you can better manage it when it happens. Consider adjusting your initial plans to account for contingencies or problems. If you face a challenge, you will have a preformed plan, and you will be less likely to be disappointed.
Learn from the situation and adjust next time.
Finally, when you experience disappointment, make sure you learn from the experience. Should you adjust your expectations next time? Was your contingency plan sufficient? As with many things, experience and practice will make you better equipped to deal with disappointment.
Disappointment is a part of life. We all experience it at one point or another. I know many have experienced what seems to be more than a fair share of disappointment in the last two years. Hopefully these tips will help you manage that disappointment.
Board of Pharmacy Update
Highlights of the September 2021 Meeting
by Matthew R. Smith, PharmD Candidate; Student Intern; University of Illinois at Chicago College of Pharmacy; Christopher W. Crank, PharmD, MS, BCPS; ICHP Executive Vice President
The September 14th Board of Pharmacy Meeting was held via conference call due to the COVID-19 pandemic. These are the highlights of that meeting.
NABP Updates: The Board gave updates on the NABP District 4 Annual Meeting. Originally scheduled as an in-person live meeting October 20-22, 2021, this meeting was switched to a virtual meeting due to the COVID-19 pandemic.
Department Updates: No Updates
Legislative Updates: IPhA Executive Director Garth Reynolds presented the legislative update to the Board. Approximately 20 bills were discussed, 18 of which became law. For more information about important legislation that was passed in Illinois, please see the Government Affairs article in this issue of
KeePosted. In addition, the details of these new laws and bills can be directly accessed through the Illinois General Assembly website at
https://www.ilga.gov/
Public Comments: During public comments, it was asked whether student pharmacists would be able to administer influenza vaccines under the supervision of other health care professionals in addition to pharmacists. IDFPR will investigate this issue and will provide the information. It was also asked if, regarding student pharmacist licensure, a college of pharmacy must report disenrollment of a student to the Department. It was stated that it is the responsibility of the student to report, but the Department encourages colleges of pharmacy to also notify the Department. IDFPR was also asked if the State of Illinois planned to sign the FDA Memorandum of Understanding Addressing Certain Distributions of Compounded Drugs. IDFPR indicated that they were aware of the MOU and that the deadline had been extended to October 27, 2022. IDFPR will provide follow up to the Board of Pharmacy on this topic.
Next Meeting: The next meeting of the Board of Pharmacy is scheduled for November 9, 2021. Interested individuals should monitor the Department’s pharmacy webpage (
www.idfpr.com) for more information. These meetings are open to the public. Pharmacists, pharmacy technicians, and pharmacy students are encouraged to attend or listen in.
Government Affairs
Division Update
Since the Illinois 102nd General Assembly started in January 2021, the State has enacted many bills affecting the practice of pharmacy. ICHP has been providing updates on the progress of these bills as well as taking an active role in advocating for certain bills, such as HB3497. Below is a summary of important healthcare legislation that has been passed in 2021.
Public Act 102-0016 (SB2017) - FY2022 Budget Implementation Act (Governor approved 6/17/2021): Updates the definition of the practice of pharmacy to include vaccination of patients 7 years of age and older pursuant to a valid prescription or standing order, by a physician licensed to practice medicine in all its branches, upon completion of appropriate training, including how to address contraindications and adverse reactions set forth by rule, with notification to the patient's physician and appropriate record retention, or pursuant to hospital pharmacy and therapeutics committee policies and procedures.
Public Act 102-0389 (HB119) - Prescription Drug Repository (Governor approved 8/16/2021): This act creates a drug repository program for the State of Illinois to which prescription drugs or supplies may be donated, provided that specified conditions are met. Drugs from the repository are considered nonsaleable, except for a reasonable handling fee. Pharmacies and pharmacists are not required to participate. Manufacturers and individuals participating in this repository are provided civil and criminal immunity.
Public Act 102-0103 (HB135) - Ins Code-Birth Control (Governor approved 7/22/2021): This act allows pharmacists to provide hormonal contraceptives to patients under a standing order from the Department of Public Health. This amends the Illinois Insurance Code to require group or individual policies to provide coverage for these pharmacist-provided services. This act is effective January 1, 2022. However, the amendment to the Illinois Insurance Code becomes effective January 1, 2023.
Public Act 102-0004 (HB158) – Health Care & Human Services (Governor approved 4/27/2021): Among various provisions, this act provides that pharmacists and pharmacy technicians must obtain at least 1 hour of continuing education in implicit bias training during their renewal period, starting in the 2022 renewal period. ICHP recognizes the need for such continuing education for the upcoming license renewal and is investigating how it may best help its members meet this CE requirement.
Public Act 102-0121 (HB279) - Drug Labeling-Gluten (Governor approved 7/23/2021): This act amends the Illinois Food, Drug and Cosmetic Act and provides that an oral drug is considered misbranded if gluten is included as an inactive ingredient and is not so listed on its label. This act does not apply to pharmacies or pharmacists.
Public Act 102-0409 (HB711) - Prior Authorization Reform Act (Governor approved 8/19/2021): This act was created with the goal of decreasing the influence of third-party payers on the patient-physician relationship, decreasing interference of third-party payers in physician decision-making, and increasing transparency of prior authorization programs.
Public Act 102-0185 (HB739) - Disease-Trichomoniasis (Governor approved 7/30/2021): This act amends the Illinois Sexually Transmitted Disease Control Act to add trichomoniasis to the list of STDs for which health care professionals may provide antibiotic treatment for the partners of infected individuals if those partners are unlikely or unable to present for comprehensive healthcare.
Public Act 102-0391 (HB1745) - Insurance-Prescription Drug Benefits (Governor approved 8/16/2021): This act amends the Managed Care Reform and Patient Rights Act. This act imposes requirements on health insurance carriers that they provide a certain number of health care plans that apply a flat-dollar copayment structure to the entire drug benefit. For individual plans, carriers must ensure at least 10% of their plans have flat-dollar copayment structure by January 1, 2023, and at least 25% by January 1, 2024. For group plans, carriers must offer at least one group health plan with this copayment structure by January 1, 2023, and at least two plans by January 1, 2024.
Public Act 102-0104 (HB3308) - Insurance-Telehealth Services (Governor approved 7/22/2021): This act amends the Illinois Insurance Code to improve insurance coverage of telehealth services. It provides that insurers shall not require that patients have prior in-person contact with the health care provider. Also, insurers shall not require proof of hardship or access barriers. Regarding reimbursement, insurers are required to cover telehealth services as they cover in-person services. Additional prior authorizations, different cost-sharing, and different reimbursement rates are prohibited by this act.
Public Act 102-0482 (HB3497) – Regulation-Tech (Governor approved 8/20/2021): This bill, now a public act, was initiated by ICHP. Before, the Illinois Board of Pharmacy did not have positions dedicated to an inpatient practicing pharmacist or a pharmacy technician, and ICHP believes that these key stakeholders in the practice of pharmacy should be represented on the Board. This law requires that one of the board members of the Board of Pharmacy be an inpatient practicing pharmacist and that one be a pharmacy technician. The number of board members is increased accordingly to 11.
Public Act 102-0490 (HB3596) - Controlled Substances-Opioids (Governor approved 8/20/2021): This act amends the Illinois Controlled Substances Act. It requires that prescriptions for Schedule II-V substances must be sent electronically, except if a prescriber certifies to the Department that he or she will not issue more than 25 such prescriptions in a 12-month period.
Public Act 102-0084 (SB194) - Pharmacy-Remote Processing (Governor approved 7/9/2021): This act amends the Pharmacy Practice Act. It provides that a student pharmacist or licensed pharmacy technician engaged in remote prescription processing of dialysate or devices necessary for home peritoneal renal dialysis at a licensed pharmacy shall be permitted to access an employer pharmacy’s database remotely while under the supervision of a pharmacist.
Public Act 102-0155 (SB579) - Facility-provided Medications (Governor approved 7/9/2021): This act became effective July 1, 2021. It requires hospitals and facilities to offer a patient any unused portion of a “facility-provided” medication upon discharge when it is administered to a patient and is necessary for continued treatment. “Facility-provided” medication is defined under Section 15.10 of the Pharmacy Practice Act as any topical antibiotic, anti-inflammatory, dilation, or glaucoma drop/ointment. Under this definition, this law applies to ophthalmic medications. This requirement only applies to medications ordered at least 24 hours in advance for surgical procedures. The pharmacist’s counseling requirement is waived, and the prescriber is required to counsel instead. Any medications must be labeled in accordance with the Pharmacy Practice Act.
Public Act 102-0400 (SB1682) - Pharmacy-Price Disclosure (Governor approved 8/16/2021): This act amends the Pharmacy Practice Act. It provides that pharmacies must disclose to the consumer at the point of sale the current pharmacy retail price for each prescription the consumer intends to purchase. Also, pharmacies must disclose if the pharmacy retail price is less than the patient’s cost-sharing amount. This act also requires that pharmacies post a notice informing customers that they may request the current usual and customary retail price for prescriptions and removes the limit to the number of disclosure requests that may be made.
Public Act 102-0527 (SB1842) - Controlled Substances-Opioids (Governor approved 8/20/2021): This act amends the Illinois Controlled Substances Act and provides that the Prescription Drug Monitoring Program may issue a report to prescribers and dispensers when a patient has 5 (previously 3) or more prescribers or 5 (previously 3) or more pharmacies that do not have a common electronic file for controlled substances in a 6-month period (previously a 30-day period).The intent of this change is to decrease the rate of false negatives identified by the Prescription Monitoring Program.
Public Act 102-0643 (SB2172) - Pharmacy Practice-Sunset (Governor approved 8/27/2021): This act provides that beginning January 1, 2024 (rather than January 1, 2022), it shall be the joint responsibility of a pharmacy and its pharmacist in charge to ensure that all new pharmacy technicians complete a standard nationally accredited training program, such as those accredited by ACPE/ASHP, or other board approved training programs.
As the practice of pharmacy evolves, it is vital that pharmacists and pharmacy technicians take an active role in advocating for the change they wish to see. To keep track of current legislation or to access current state laws, one may visit the ICHP advocacy webpage or the Illinois General Assembly website (
https://www.ilga.gov/).
Professional Affairs
2021 ASHP House of Delegates Update
by Andrew Donnelly, PharmD, MBA, FASHP; Director of Pharmacy - University of Illinois Hospital, Associate Dean for Clinical Affairs -University of Illinois at Chicago College of Pharmacy; Noelle RM Chapman, PharmD, BCPS, FASHP; VP Pharmacy Operations, Pharmacy - AdvocateAuroraHealth; Bernice Y. Man, PharmD, BCPS, CSP Practice Coordinator, Specialty Pharmacy - Northwestern Medicine; Carrie Vogler, PharmD, BCPS Clinical Associate Professor - SIUE School of Pharmacy
In June 2021, ASHP House of Delegates met virtually. The following table gives a brief overview of the approved policies. The full version of policies under consideration and ASHP member comments can be found on the ASHP Connect website. The following statements were also approved: Statement on the Roles and Responsibilities of the Pharmacy Executive, Statement on the Pharmacist's Role in Public Health, and Statement on the Pharmacist's Role in Clinical Pharmacogenomics. A new business item COVID-19 Vaccination Requirements to Advance Patient Safety and Public Health was sponsored and presented by Bernice Man, ICHP member, and was well supported by the House.
Thanks to all the ICHP members who provided feedback on the policies before the meeting. Illinois delegates took your recommendations into consideration when representing our organizational membership. We look forward to deliberations of the next House of Delegates next June at the 2022 ASHP Summer Meeting in Phoenix, AZ.
Professional Affairs
Attention ASHP Pharmacist Members - It's Time to Elect Delegates to ASHP's House
by Christopher W. Crank, PharmD, MS, BCPS; ICHP Executive Vice President
The ASHP Summer Meeting for 2022 will be held in Phoenix, Arizona from June 11-15. ASHP’s House of Delegates meets during the Summer Meeting. ICHP needs to elect two delegates to join Noelle Chapman, Andy Donnelly, and Carrie Vogler as the Illinois delegation for 2022 and then continue in 2023. ASHP’s House of Delegates is the policy making body within ASHP and is responsible for approving all ASHP position statements and practice guidelines.
Delegate candidates must be ASHP pharmacist members who plan on attending the ASHP Summer Meetings for both 2022 and 2023 at their own expense. In addition, they must attend one of the Chicago-based Regional Delegate Conferences each May.
Nominees must provide nomination statements (limited to 250 words or less), which include years of membership in ASHP, current employment position, pharmacy association memberships, volunteer experiences related to pharmacy associations, and any other relevant information the potential candidate would like to include. The ICHP Committee on Nominations will select up to six candidates for this year’s ballot. The two candidates receiving the highest vote totals will be credentialed as delegates for 2022 and 2023 and the remaining candidates will serve as alternate delegates for 2022 only.
This is a great opportunity for someone who has served at the state level for some time and wishes to move up within ASHP. It is probably not a good match for someone with no pharmacy association volunteer experience.
Elections will be held at the end of November, with all ASHP pharmacist members eligible to vote on-line. Election results will be announced in the February
KeePosted. Interested individuals should send their letter of intent and candidate’s statement to Chris Crank by November 15th at the ICHP office via fax at (815) 227-9294 or e-mail to
chrisc@ichpnet.org.
Professional Affairs
The Pharmacist's Role in Promoting Vaccine Acceptance
by Karen Davidge, PharmD PGY2 Infections Diseases Pharmacy Resident Advocate Lutheran General Hospital; Mary Palmer, PharmD, BCPS PGY2 Infectious Diseases Pharmacy Resident Advocate Lutheran General Hospital; Jen Phillips, PharmD, BCPS, FCCP, FASHP Professor of Pharmacy Midwestern University College of Pharmacy, Downers Grove
According to the World Health Organization (WHO), vaccine hesitancy - defined as the reluctance or refusal to vaccinate despite the availability of vaccines - is considered one of the top ten threats to global health.1 In a systematic review of vaccine acceptance rates worldwide completed in December 2020, a range from 23.6% to 97% of people reported they would be willing to receive a COVID-19 vaccine.2 Surprisingly, the United States is among those countries with lower vaccination acceptance rates, with 56.9% of the population willing to receive a COVID-19 vaccine if given the opportunity. A recent survey in April 2021 assessing vaccine hesitancy among the U.S. population revealed that hesitancy rates vary widely across the U.S., ranging from as low as 4.5% to up to 25% of the population living within a specific geographic area. In Illinois specifically, between 6.8% to 9.5% of the population reported having some degree of hesitancy regarding the COVID-19 vaccine in April 2021. Fortunately, overall rates of vaccine hesitancy in the U.S. have been declining in recent months. Per the Centers for Disease Control and Prevention (CDC), predictors of higher vaccine hesitancy rates include female gender, age 25-39 years old, no college degree, lower income, and being uninsured or having Medicaid. The lowest vaccine hesitancy rates are among those aged 65 years or older and of Asian (non-Hispanic) descent. Rates of vaccine hesitancy tend to be lowest on the West Coast and in the Northeast, and highest in the South, Great Plains, and Alaska.3
Given such high rates of COVID-19 vaccine hesitancy, both in the U.S. and worldwide, it is vital to identify and address factors contributing to vaccine hesitancy. Some of the most commonly cited reasons for vaccine hesitancy include concerns about the safety profile of the COVID-19 vaccines and mistrust of the government or other organizations that recommend mass vaccination programs. Individuals may also be reluctant to get vaccinated if they believe they are at low risk of harm due to COVID-19 infection. An online survey of U.S. residents conducted in September 2020 found that 68.6% of respondents planned to be vaccinated against COVID-19 once a vaccine became available. However, most respondents (63.5%) said they were worried about the possible side effects of a COVID-19 vaccine, indicating this is a concern even for those who are willing to accept the vaccine. This was likely related to the perception that the vaccine testing and approval process was occurring too quickly, as 73.1% of respondents expressed concern that “the rushed pace for testing for a new COVID-19 vaccine will fail to detect potential side effects or dangers.” When presented with three different hypothetical levels of efficacy (50%, 75%, or 99% protection against COVID-19 infection), respondents were significantly more likely to indicate a willingness to be vaccinated if the vaccine's efficacy was higher.4 Similar results were reported in a survey of Medicare beneficiaries conducted in October and November 2020. The most common reasons for unwillingness to accept a COVID-19 vaccine were “not trusting government” (42.1%), “side effects” (41.3%), “the vaccine not preventing COVID-19” (11.3%), and “the vaccine causing COVID-19” (10.1%).5 These survey results suggest that educational campaigns to promote uptake of the COVID-19 vaccines should emphasize the high efficacy and low rates of severe adverse effects associated with the COVID-19 vaccines while also outlining the strict requirements for vaccine approval and post-marketing monitoring of vaccine safety.
A large randomized controlled trial performed in the United Kingdom during January and February 2021 measured the impact of different educational interventions on willingness to be vaccinated among individuals who endorsed COVID-19 vaccine hesitancy. The investigators determined that for strongly hesitant study participants, providing information on the personal benefits of vaccination had a higher impact compared to providing information on the collective benefits of vaccination (or providing information on both personal and collective benefits).6 A recent commentary on evidence-based strategies for addressing COVID-19 vaccine hesitancy proposed that clinicians should tailor their vaccine recommendations to better connect with the priorities and concerns of each patient. The authors recommend that healthcare providers correct vaccine misperceptions by (1) affirming the patient’s personal values or worldview, (2) explaining the motivation behind vaccine misinformation, and (3) repeating factual information about the vaccines.7
As one of the most accessible healthcare providers, the pharmacist is pivotal to help reduce vaccine hesitancy and help guide patients towards vaccine acceptance. Changing the terminology from “vaccine hesitancy” to “vaccine acceptance” to help frame the issue in a more positive perspective is one way to do this. Other strategies are outlined below.8,9
Prevent/Correct Misinformation. There is an abundance of information available to consumers and patients related to vaccines. Some sources provide helpful and objective information, whereas others contain “myths” or misinformation. Pharmacists can help steer patients towards more reputable sources of information. Websites, social media, and apps with content written by the government or scientific agencies or by credible experts are more likely to contain accurate information. Other sources such as blogs, social media posts, and unreferenced content may not be accurate or up to date. Did you know that the FDA has vaccine information available in more than 20 different languages? https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/multilingual-covid-19-resources
Use Two-Sided Conversations. It is essential to listen to the patients' concerns. By asking open-ended questions, pharmacists can determine the primary barrier to getting vaccinated. Instead of asking, “Why haven’t you received your vaccine yet?” try “What are your thoughts about the COVID-19 vaccine?” Acknowledge fears and concerns, but steer the conversation back towards objective, credible evidence. Using motivational interviewing concepts, you can also tailor the message you provide based on the specific reason(s) acknowledged for vaccine hesitancy.
Have a Balanced Discussion. When discussing the vaccine with patients, the conversation should include objective information on both efficacy and side effects. Be honest about what is known and what is unknown. Do not “oversell” the benefits or “undersell” the risks.
Keep Current. Find a way to keep up to date on new information and treatment strategies that become available. Also, understanding the latest “misinformation” might help you gather evidence to dispute unfounded claims.
Champion the Issue. One critical way pharmacists can help advance the COVID-19 vaccine initiative is to lead by example. If you’ve received your vaccine already, let people know. Some workplaces offer stickers or buttons to wear. You can also use social media to help propel the cause. Let your friends and followers know that you are available as a resource to answer any questions and post credible information on your social media pages/apps.
Vaccine hesitancy is not unique to COVID-19. What’s more, people are generally not on “one side” or the other. Acceptance of vaccination is best considered in terms of a continuum. Some refuse all vaccines, others accept all, and many may refuse or accept, but with some concerns. Pharmacists are in a great position to serve as an objective resource on COVID-19 and other vaccines. Hopefully, our profession can help educate the public on vaccines and serve as a valuable resource in the process by utilizing some of the strategies outlined above.
References
- World Health Organization. Ten threats to global health in 2019. Available at: https://www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019. Accessed 20 Jun 2021. Accessed 17 Jun 2021.
- Sallam M, Gori D. COVID-19 vaccine hesitancy worldwide: a concise systematic review of vaccine acceptance rates. Vaccines (Basel). 2021;9(2):160. Doi: 10.3390/vaccines9020160.
- Centers for Disease Control and Prevention. Estimates of vaccine hesitancy for COVID-19. Available at: https://data.cdc.gov/stories/s/Vaccine-Hesitancy-for-COVID-19/cnd2-a6zw/. Accessed 17 Jun 2021.
- Pogue K, Jensen JL, Stancil CK, et al. Influences on attitudes regarding potential COVID-19 vaccination in the United States. Vaccines (Basel). 2020;8(4):582. Doi: 10.3390/vaccines8040582.
- Luo H, Qu H, Basu R, et al. Willingness to get a COVID-19 vaccine and reasons for hesitancy among Medicare beneficiaries: results from a national survey. J Public Health Manag Pract. 2021; epub ahead of print. Doi: 10.1097/PHH.0000000000001394.
- Freeman D, Loe BS, Yu LM, et al. Effects of different types of written vaccination information on COVID-19 vaccine hesitancy in the UK (OCEANS-III): a single-blind, parallel-group, randomised controlled trial. Lancet Public Health. 2021;6(6):e416-e427. Doi: 10.1016/S2468-2667(21)00096-7.
- Rutten LJF, Zhu X, Leppin AL, et al. Evidence-based strategies for clinical organizations to address COVID-19 vaccine hesitancy. Mayo Clin Proc. 2021;96(3):699-707. Doi: 10.1016/j.mayocp.2020.12.024.
- Terrie YC. The role of the pharmacist in overcoming vaccine hesitancy. US Pharm. 2021;45(4):28-31.
- Banner L. COVID-19 provides opportunity to rethink vaccine hesitancy. Innovations. 2021;27(5):26-31. Doi: 10.1016/j.ptdy.2021.04.011.
Professional Affairs - Best Practice Award Winner!
Software Aided Compounding: Adopting Technology to Improve Patient Safety and Documentation
by By Ron Hartman, PharmD - Regional Director of Pharmacy Operations - OSF Healthcare - Galesburg, IL; Heather Harper, PharmD, BCPS - Pharmacy Informatics Specialist - OSF Healthcare - Peoria, IL ; Elizabeth Harthan, PharmD, BCPS - Anticoagulation Pharmacotherapist Coordinator - OSF HealthCare Ministry Services - Peoria, IL ; Patricia M. Kohli, BSPharm - Pharmacy Operations Manager - OSF St. Francis Hospital & Medical Group - Escanaba, MI; Jamie Martin, PharmD - Regional Informatics Pharmacist - OSF Healthcare - Peoria, IL ; Karin L. Terry, PharmD, CPPS - Medication Safety Pharmacist OSF Healthcare - Peoria, IL

Introduction, Purpose, and Goals of the Program
Compounding is an essential function of health system pharmacy practice, and achieving precision in compounding is vital to patient safety. As with most areas of pharmacy practice, a close eye for detail could be the difference between success and disaster. The Institute for Safe Medication Practices shed light on this when they reported on a case of a child who died when an oral product was compounded in error.1 The wrong active ingredient was incorporated into the compound, and a final check was either delayed or the ingredients were misidentified.
The American Society of Health-System Pharmacists (ASHP) has also addressed the importance of introducing barcode scanning in compounding.2 In their position statement, they support barcode scanning in compounding to enhance patient safety and quality of care by improving the accuracy of this core pharmacy function. They also detail how an audit trail can be created from barcode scanning. This trail can be used to track and trace a compounded product from pharmacy to patient.
The purpose of our project is to standardize compounding across our health system. By standardizing formulas for sterile and non-sterile compounds, and adding those formulas into the electronic health record (EHR), we are able to utilize ingredient barcode scanning to prevent tragic errors like the one highlighted here, and align with ASHP guidance. This process will not only prevent errors, it will also be used to link the ingredient lots and expiration dates to the patient record, creating the described audit trail, and making retrieval much less difficult in the event of a recall or compounding error.
The goals for this project are: standardize compounding processes across a multi-site health system with facilities ranging from a large medical center to critical access hospitals; build standardized compounding formulas into the EHR software; use barcode scanning during compounding to identify ingredients and document pharmacist verification; and reach 80% compliance with this process for all facilities within the health system.
Description of the Program
Our health system uses Epic® as the EHR. It has activities called Compounding and Repackaging (CNR) and Dispense Prep, which can be used to accomplish the goals of this project. These activities require configuration, but are included in our software system at no additional cost. They can be incorporated without the time or expense required to procure an outside software vendor. We convened a team of pharmacists and technicians from across our health system that worked together to compile a list of commonly-used compounds. We standardized the compounding procedure for each formula and assigned common Beyond Use Dates (BUDs), using primary literature references when possible, or referencing USP <797> and <795> when not available.
Once the formulas and BUDs were finalized, we consulted with our IT pharmacy support team to build the compounds in the EHR, ensuring procedures and functionality could adapt to the needs of our facilities of varying scope and size. Both CNR and Dispense Prep allow for flexibility in vial size used during compounding—one facility could use a 1g vial of vancomycin, for instance, while another could use a 10g vial.
During compounding, pharmacy staff accesses the CNR and Dispense Prep activities within the Epic® Willow module. Dispense Prep is intended for use in patient-specific compounding, and will accordingly link the ingredient lots and expirations to the final dispensing on the patient’s EHR. CNR is intended for batch compounding, and will link component lot number and expiration information to a patient when a nurse scans the medication at bedside.
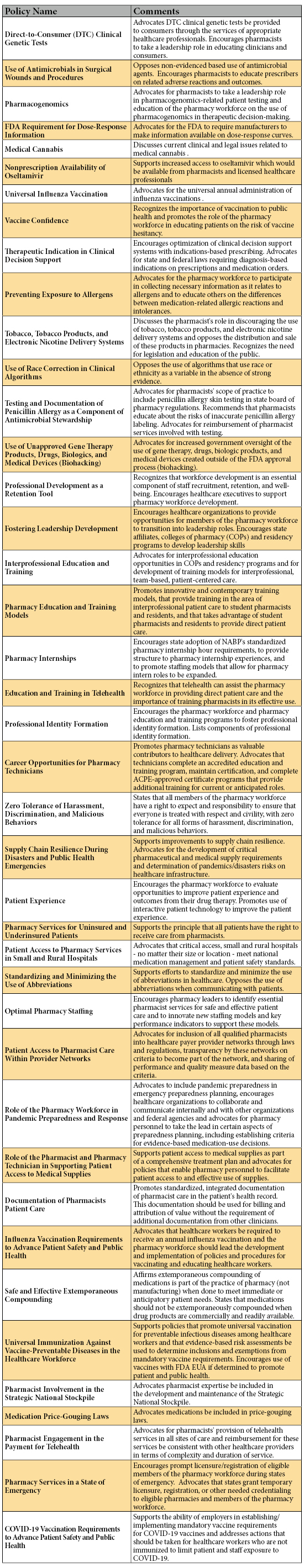
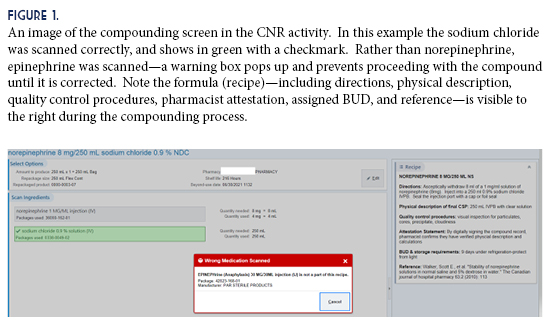
To produce a batch compound using CNR, pharmacy staff selects the desired CNR record to display the formula and ingredients (Figure 1). If an ingredient is incorrectly scanned, a warning message will display, and cannot be bypassed until it is rectified. Once the compound is completed, the verifying pharmacist uses another activity called Compounding and Repackaging Check. Here the label barcode is scanned again and a summary screen will display the ingredients used, as well as any errors that occurred (Figure 2). When the pharmacist is satisfied with the compounding accuracy, they can verify and approve the compound. If errors cannot be explained during the review, the pharmacist can also reject the compound, in which case it would route back to be compounded again. The pharmacist uses a similar activity called Dispense Check to verify patient-specific doses using Dispense Prep.
Experience with Outcomes of the Program
We almost immediately realized how complex standardizing compounding would be. In addition to internal capabilities, Epic® also receives information from Medi-Span®, our medication data vendor. The way our health system uses Medi-Span®’s classifications of diluents can limit their interchangeability. To continue the example used earlier, we can use different vial sizes of vancomycin, but we cannot switch between sterile water for injection vials and bags—vials are categorized in Medi-Span® as injections (IJ SOLN), and bags are listed as infusions (IV SOLN). This meant we needed to further standardize, not only to formula and BUD, but also to which presentation of diluent was used throughout the health system.
Once Dispense Prep and CNR were rolled out across the system, they were widely accepted. Electronically capturing our compounding efforts meant we no longer had to keep often lengthy paper logs to comply with regulatory requirements. It also increased our productivity and reproducibility—the need to hand-write documentation was eliminated, and the ability to electronically pull up the record if needed prevented a laborious search, both saving time. In the first six months of Fiscal Year 2021, system-wide compliance with Dispense Prep has reached 64.6%, with a goal to reach 80% compliance by Fiscal Year-end.
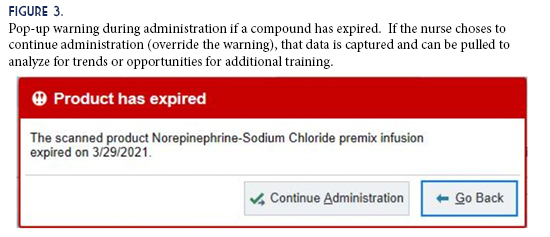
An unexpected benefit from the project was an added layer of protection against administering expired compounds. Since the formulas and BUDs are built into the EHR, the software can track expiration dates. If a compound is expired, nursing staff is alerted via a pop-up message during administration (Figure 3). This warning can be overridden, but it does add another tier of safety that may prevent administration errors potentially leading to patient harm. Overriding a warning generates trackable data that can be pulled and analyzed for trends or opportunities for additional training.
Studies have shown that barcode scanning can result in a relative reduction in the incidence of errors by 93-96%.3 This is particularly impactful in compounding, where errors are difficult to detect once they leave the pharmacy. Prior to our project, verification of compounding was an entirely human process. Implementing barcode scanning and standardized compounding formulas allow us to be more confident in our ability to identify compounding errors before they reach the patient. Additionally, our IT pharmacy support team was able to use captured data to generate a Near Misses dashboard (Figure 4). This dashboard allows us to review software-discovered issues in compounding, to identify trends and reinforce training where needed.

Another benefit of the program is the pass-through of drug NDC numbers, from wholesaler ordering to patient dispensing. A mismatch of NDC numbers, where one is dispensed and another is documented or billed, is the definition of misbranding. While this is prohibited in the Food, Drug, and Cosmetic Act, accuracy of billed NDC numbers is also especially important for facilities that participate in the HRSA 340B Drug Pricing Program. Where applicable, facilities are granted accumulations of drugs at a discounted rate, based on use in low-income or at-risk patient populations. Verifying NDC pass-through ensures compliance with the HRSA program, improves regulatory compliance, and maintains health system cost-savings from that program. The compounding project enables our health system to track and trace NDC pass-through, which validates our compliance with the 340B Drug Pricing Program requirements.
Discussion of Innovative Aspects of Program and
Achievement of Goals
This project proved to be an innovative and far-reaching application at our health system. Not only did it meet the initial goal of identifying and preventing the dispensing of compounds made in error, it also improved productivity of pharmacy staff by eliminating paper documentation, aided in validation of our 340B program, and added another layer of awareness to nursing staff to prevent accidental administration of expired compounds.
One of the major undertakings of this project was to develop standardized compounding formulas, and have them built within the EHR. This ensured that all sites were following the same procedure, resulting in a consistent patient and healthcare team experience, no matter where that person was within the health system. Previously we relied on paper formulas that could be easily lost or become obsolete, or could be misinterpreted when transcribed from one location to another. Now our EHR is the source of truth, eliminating many of those duplicative and time-consuming risks.
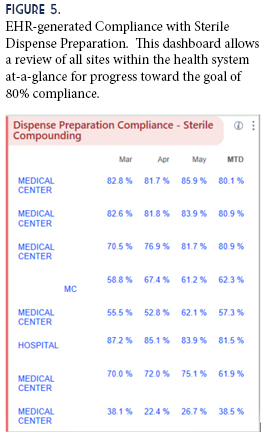
Another innovative aspect was the development of metrics to allow us to consistently measure our compliance with the Epic® activities. Previously we did not have an easily-measurable metric to validate compounding compliance. Now we can pull compliance data from our EHR, comparing all facilities within the health system for Dispense Prep (Figure 5). This filled a gap in Quality, Safety, and Regulatory oversight, and satisfies many of the questions in this area that are frequently raised during inspections or surveys. Development of the metrics led to a new standard for compliance—the goal of 80% use of Dispense Prep by Fiscal Year-end. This goal has yet to be reached, and is ongoing.
Conclusion
Utilizing software-aided compounding, through barcode scanning and documentation, has been a great success for our health system. Standardized formulas, built in the EHR, ensure that all facilities are consistently producing compounds with the same process. Adopting BUDs that flow through the EHR provides added protection against administration of expired compounds. Ingredient barcode scanning adds greater oversight, where a pharmacist can see at-a glance if the compound was prepared correctly, and has led to an increased confidence in the accuracy of our compounded products. The pass-through of drug NDC numbers to patient administration provides additional benefits, preventing misbranding and preserving beneficial purchasing through government programs. Finally, being able to utilize activities that already exist in our EHR allowed us to make these gains in safety and accuracy without a large capital investment.
Our experience has shown that, while there is considerable investment in time to develop a program utilizing software aided compounding, the benefits are numerous, to both the health system and the patients we serve. Some of the positive impacts were not even apparent until after we started the project. We would encourage any health system to investigate the possibilities within their own EHR to do the same.
References
- Institute of Safe Medication Practices. Death Due to Pharmacy Compounding Error Reinforces Need for Safety Focus. June 15, 2017. Available at Death Due to Pharmacy Compounding Error Reinforces Need for Safety Focus | Institute For Safe Medication Practices (ismp.org) Accessed June 21, 2021.
- American Society of Health System Pharmacists. ASHP statement on barcode verification during inventory, preparation, and dispensing of medications. Am J Health-Syst Pharm. 2011; 68-442-5
- Poon EG, Cina JL, Churchill W, Patel N, et al. Medication dispensing errors and potential adverse drug events before and after implementing bar code technology in the pharmacy. Ann Internal Med. 2006 Sep 19; 145(6): 426-34

New Practitioners Network
Building a Goal-Oriented Career
Looking into the future of one’s career can be both exciting and intimidating, with endless opportunities and numerous paths waiting to be explored. In such a large expanse of options, goal setting can add structure and facilitate progress by focusing and outlining a plan to move forward.
As I sat down to begin my first job, no longer a trainee, I was vexed with this new sense of career ambiguity. After working within structured curriculums and requirements for so long, I wondered, “What are the next steps in building my career? How do I proceed?” So, as I sat down on my first day, I began to write down my goals for the upcoming year.
In my previous work and observations as a PGY2 Health-System Pharmacy Administration and Leadership Resident, goal setting was a key component of planning at all levels of the organization. Institutionally, annual goals are presented by executive leadership, outlining how the organization plans to grow and improve over the next few years. From a departmental level, strategic planning is performed every few years to update the mission, vision, and strategic goals of pharmacy practice. Within pharmacy services, smaller teams (e.g., project groups) may utilize quarterly or annual goals to unite members under a common purpose and prioritize their initiatives.
Goals establish a shared vision, set benchmarks and metrics to track progress, maintain focus, and build teamwork between group members. The same benefits apply to individual goals, with the additional advantage of being able to account for a holistic perspective, including personal wellness and non-career responsibilities and passions.
Getting started:1,2
- Write out your goals:
- The format of goal-writing should be tailored to individual preferences. Some ideas include: separating goals into major and minor goals, sorting your goals by category (e.g., career, school, professional organizations, hobbies, health), or organizing goals by a specific value (e.g., organizational impact, personal meaningfulness).
- Ensure a mix of short-term and long-term goals. Long-term goals can create a drive for future gratification, but short-term goals are equally important to maintain a sense of accomplishment as you achieve these milestones.
- Account for non-career goals, especially if you spend significant time pursuing hobbies or studies outside of pharmacy.
- Define a timeframe:
- Be realistic about your timeframe and the amount of work you can manage.
- Long-term goals may need to be continuously reassessed.
- Create measurable goals:
- Set metrics to know when you have achieved your goal. This could be a numerical value, a qualitative measure, or an action to be completed.
Once composed, review the list of goals to assess the overall balance, manageability, and value these goals add to one’s career and life. For example, in the article, “Stop Setting Goals You Don’t Actually Care About” written by Elizabeth Grace Saunders, she encourages people to ask these three questions when setting goals:2
- If you could accomplish just one major professional development goal, what would it be?
- When you think about working on this goal, do you get excited about both the process and the outcome?
- Is your motivation to pursue this goal intrinsic because it is personally interesting and important, or is it extrinsic, which you feel would please other people?
These questions encourage reflection to pick goals that align with what matters to an individual, ensuring one’s motivations are based on internal desires, passion, and personal vision rather than external expectations.
Taking time to determine one’s goals is highly beneficial, especially at the start of one’s career when there are so many options and opportunities to invest time and effort. Goals will continue to evolve as one faces new experiences, connections, and challenges. Though it may seem challenging to control something that is ever-changing, there are certain steps one can take to ensure goal setting is a productive and successful process.
Maintaining and reaching your goals:1
- Set check-ins to hold yourself accountable:
- Schedule a time (e.g., bi-weekly, monthly, quarterly) to review and update your goals and measure progress.
- Consider a public commitment - maintain accountability for your progress by regularly reviewing goals with a peer or mentor.
- Reassess your goals whenever you need to:
- Responsibilities can continually evolve, especially following unforeseen events (e.g., a pandemic) or significant changes (e.g., moving, children), shifting priorities, and sometimes even your career direction.
- Find support, such as a mentor or preceptor, to help you reach your goals, discuss how to overcome challenges, or connect you with potential opportunities.
- Keep in mind that not reaching your goals is not a failure - it is an opportunity to reassess:
- If only a few goals are unmet by their original timeframe, it is possible that specific obstacles need to be addressed.
- If many goals are left unmet, reprioritize your goals (including putting some on the back burner), set a more realistic timeframe, and create smaller action items to dissect lofty goals into feasible steps.
- Maintain ongoing records of your progress. Utilizing your goal-setting documentation to keep track of the work and projects you have completed will be helpful to your future curriculum vitae (CV) writing and interview discussions when needing to recall your past experiences.
No matter where one’s pharmacy journey may lead, honing the skill of goal setting allows both new and experienced professionals to take charge of their career planning and build a sustainable and fulfilling career.
References:
- Saunders EG. Stop Setting Goals You Don’t Actually Care About. Harvard Business Review. https://hbr.org/2016/12/stop-setting-goals-you-dont-actually-care-about?referral=00203&utm_source=newsletter_management_tip&utm_medium=email&utm_campaign=tip_date. Published May 3, 2017.
- Grote, D. 3 Popular Goal-Setting Techniques Managers Should Avoid. Harvard Business Review. https://hbr.org/2017/01/3-popular-goal-setting-techniques-managers-should-avoid. Published January 2, 2017.
- Greenhaus JH, Callanan GA, Kaplan E. The Role of Goal Setting in Career Management. International Journal of Career Management. 1995;7(5):3-12. doi:10.1108/09556219510093285.
Educational Affairs
Illinois Council of Health-System Pharmacists Poster and Platform Presentations for Spring Meeting
ICHP POSTER CATEGORIES:
The following are the categories for submission:
Original Research:
To be considered for acceptance, papers must describe original research in pharmacotherapeutics, clinical pharmacokinetics, pharmacodynamics, or practice innovations in organized health-care settings. Additionally, administrative practices,cost analyses, or clinical/quality outcomes of pharmaceutical services and scholarship of teaching and learning submissions may also be accepted. Papers must not have been published in abstract or complete form or have been presented elsewhere prior to presentation at the ICHP Spring Meeting.
Research in progress:
To be considered for acceptance, papers must describe original research in pharmacotherapeutics, clinical pharmacokinetics, pharmacodynamics, or practice innovations in organized health-care settings. Additionally, administrative practices,cost analyses, or clinical/quality outcomes of pharmaceutical services and scholarship of teaching and learning submissions may also be accepted. Papers must neither have been published in abstract or complete form nor have been presented elsewhere prior to presentation at the ICHP Spring Meeting. Those abstracts submitted as research in progress are expected to have results and conclusions available for poster presentation at the Spring Meeting. Those with no results and conclusions by the time of the meeting are disqualified.
Encore:
Research may have been presented elsewhere or published in abstract or complete form only prior to presentation at the ICHP Spring Meeting. To be considered for acceptance, papers must describe original research in pharmacotherapeutics, clinical pharmacokinetics, pharmacodynamics, or practice innovations in organized health-care settings. Additionally, administrative practices,cost analyses, or clinical/quality outcomes of pharmaceutical services and scholarship of teaching and learning submissions may also be accepted. Posters or papers presented before January 1, 2021 are not eligible. Full results and conclusions must have been presented/published previously to qualify as an encore presentation. Those projects previously presented/published without results and conclusions should be submitted as an original poster.
Student:
Only original material will be considered. Any material that has already been presented should be entered as an encore presentation. The person primarily responsible for the work must be a current student enrolled in an Illinois School of Pharmacy and must present the poster at the meeting.
Original research may be submitted as research in progress. Those abstracts submitted as research in progress are expected to have results and conclusions available for poster presentation at the Spring Meeting. Those with no results and conclusions by the time of the meeting are disqualified.
Award-Ineligible Category:
Case Report/Series: A detailed report of the diagnosis, treatment and follow-up of an individual patient or small group of patients, usually describing an unusual or novel occurrence. These reports will not be eligible for poster judging or awards.
**Literature reviews (excluding meta-analyses) are not eligible for poster submission. This type of research may be eligible for submission to ICHP’s News journal, KeePosted.
PLATFORM PRESENTATIONS: Abstracts submitted with results and conclusions will be eligible for a platform presentation (excluding case reports/series), as decided by the ICHP Educational Affairs Division Poster Review Committee. In addition to a research abstract, individuals submitting an abstract with results and conclusions must also submit one learning objective that: 1) is encompassing of the overall project, and 2) uses one of the approved knowledge-based verbs listed below:
- Arrange
- Classify
- Define
- Describe
- Discuss
- Duplicate
- Explain
- Express
- Identify
- Indicate
- Label
- List
- Locate
- Memorize
- Name
- Order
- Outline
- Recognize
- Relate
- Recall
- Repeat
- Reproduce
- Report
- Re-state
- State
For further information about the platform presentation, see the Review of Submissions section below.
ELIGIBILITY CRITERIA: All Illinois health-system pharmacy practitioners, including pharmacists, pharmacy residents, pharmacy technicians, and pharmacy students are invited to submit projects for consideration. The submitting author must be a current ICHP member. If a student is listed as the submitting author for an original presentation, the poster will be eligible for the student award.
The submitting author should present the poster; however, if legitimate circumstances do not permit attendance at the meeting; another pharmacy professional author for that poster may present at the poster during the poster exhibit session at the Spring Meeting. All poster presenters must be registered for the Spring Meeting and complete ACPE required documents as requested if the posters are accredited for continuing pharmacy education.
SUBMISSION OF POSTERS: Individuals wishing to present a poster should submit the information through the ICHP website: https://ichpnet.org/pharmacy_practice/pharmacy_education_and_cpe/posters/poster_guidelines.php.
REVIEW OF SUBMISSIONS: All submissions from eligible authors received by the designated deadline will be reviewed anonymously by the Educational Affairs Division Poster Review Committee for their acceptability for presentation. Submissions will be reviewed for their originality and applicability to health-system pharmacy and scored on relevance/originality, objectives, methods, results and conclusions/implications. See poster guideline page for abstract scoring sheet.
In order for an original or encore submission or a case report to be accepted for poster presentation, a minimum score of 5 on the abstract scoring sheet is required. Research in progress requires a minimum score of 3, as results and conclusions/implications will not be scored. Two blinded reviewers must score the abstracts at the minimum required score in order for it to be accepted for poster presentation at the Spring Meeting. In the case of a split decision between the 2 blinded reviewers, a 3rd blinded reviewer will score the abstract and the abstract will be accepted if the minimum required score is also given by the 3rd reviewer.
The two abstracts in the original research categories with the highest scores (minimum of 7), submitted with results and conclusions at the time of abstract submission, will be invited to participate in the platform presentation session at the Spring Meeting. If there are none that qualify, encore research with results and conclusions will be considered if it receives a score of 7 or higher. Each presentation will be 15 minutes in length. These presenters will receive a platform presentation award (see Awards section below). If a presenter opts out of the platform session, they will still be eligible for a poster award. The next highest score will be invited to participate in the platform session if a presenter opts out of the platform session. Those accepted for a platform presentation will be asked to provide additional information for ACPE accreditation purposes. Three judges will be selected to score the presentations.
The five abstracts with the next highest scores from the Original Research and Research In Progress categories combined will be eligible for the “Original” poster awards. The five abstracts with the top highest scores in the Encore and Student categories will be eligible for the “Encore” and “Student” poster awards, respectively. A project presented during the platform session is not eligible for a poster award and vice versa.
Although all posters received by the deadline will be reviewed on their merits, in some instances, due to space limitations, only a limited number of posters will be accepted for presentation at the meeting. For this reason, authors of poster submissions are encouraged to submit their entries early since, under these circumstances, posters will be accepted for presentation on a “first come, first served” basis.
All submitting authors will be notified of the acceptance of their poster as either a platform presentation or a poster presentation via e-mail. Authors whose posters are rejected will be provided with a detailed explanation of the committee’s reason(s) for their decision.
AWARDS:
Best Platform Presentation – Recognition plaque and winner’s pharmacy department will receive one free ICHP state-wide full meeting registration of their choice to give to one employee.
Best Original Poster – Recognition certificate and the winner’s pharmacy department will receive 50% off one ICHP state-wide full meeting registration of their choice for one of their employees.
Best Encore Poster -
Recognition certificate
Best Student Poster -
Recognition certificate and the winner’s ICHP student chapter will receive one free ICHP state-wide full student meeting registration of their choice for one of their members.
Posters/platform presentations will be judged at the ICHP Spring Meeting, during which time the winners will be announced.
Submissions due by January 10, 2022
ICHP Leadership Spotlight
Meet Erika Diericx, PharmD, BCPS
What is your ICHP leadership position?
Southern Region Director
Where is your practice site?
Southern Illinois Healthcare (SIH) – System ID/ASP Clinical Pharmacist
What is a challenge that you face in your practice?
I think the biggest obstacle I face is engaging others to embrace change. Sometimes, as pharmacists, we are happily set in our ways of practice – I think most pharmacists I know are very much creatures of habit. I am a creature of habit. But I think sometimes we as a profession let the fear of change get in the way of the change itself.
What have you done to improve a problem at your organization?
To aid in earlier Infectious Diseases consults I lead the implementation of an alert within our electronic medical record (Epic – BPA) to recommended an Infectious Diseases consult to the patient’s primary team for assistance in treatment of all S. aureus bacteremia and any fungemia.
What makes ICHP great?
I think the opportunities ICHP provides is what makes ICHP great – from the bi-annual meetings to the leadership opportunities for pharmacists, pharmacy students, and pharmacy technicians. I serve a rural community in a healthcare organization that does it's best to provide cutting edge services. I find it helpful to be able to have a network of pharmacists within reach to further develop my own team.
What initially motivated you to get involved in ICHP?
I was introduced to ICHP through a leader and mentor - Jennifer Ellison. I was a newly hired pharmacist at OSF in Peoria. I was astonished that a member of pharmacy leadership had taken time to discuss opportunities for networking and clinical growth. My positive perception of ICHP began that day – I wasn’t just a member who would get lost in the numbers and dues.
Is there an individual that you admire or look up to, or a mentor that has influenced your career?
Each phase of my career has had many people that contributed to the way I practice pharmacy. When I was in community pharmacy my mentor Mark laid the groundwork. His gentle nudge towards an inpatient clinical pharmacist role is what planted the seed. It was the pharmacists at SFMC that nourished it to grow. I will always be grateful for the team who developed my skills as an inpatient pharmacist.
What advice would you give to student pharmacists?
My advice would be to remember your passion and this will drive your success. One of my passions is learning. I have made a few career changes in the pursuit of knowledge. If you want something and put your mind to it, you can achieve it. For me, in my pursuit of knowledge, career opportunities have presented themselves. Find what makes you passionate about pharmacy and pursue it.
Do you have any special interests or hobbies outside of work?
Pre-COVID I loved to travel. When my daughter was born, I was excited to have her come along for the adventure. While the pandemic has shaped our travel experiences differently than I anticipated we have still made the best of it.
Where is your favorite place to vacation?
My favorite place to vacation is London. I loved how easy it is to travel within England and throughout Europe. My husband is an avid Chelsea FC fan – seeing them in a match against Arsenal was amazing. We also visited the many historical sites and shopping!
ICHP Leadership Spotlight
Meet Russell Jason Orr, PharmD, MBA
My Story:
I am currently the Executive Director of Inpatient Pharmacy Services at the University of Chicago Medicine however my journey to this point may not be what one would expect. My career to date has spanned being a retail Pharmacy Technician, a hospital Pharmacy Technician, a Pharmacy Student, a Pharmacy Resident, a pediatric Clinical Pharmacist, a pediatric Operational and Clinical Manager, a pediatric Clinical Director, and a children’s hospital Pharmacy Director. These experiences have allowed me the exposure to understand how intricate and powerful our profession is.
What is your ICHP leadership position?
I am the Northern Region Director and am excited to get involved with ICHP at a higher level.
Where is your practice site?
University of Chicago Medicine in Hyde Park
How would you change pharmacy if you could?
Every leader may say “unlimited resources” would change pharmacy but every leader would also say “come back down to reality!” What remains a reach for our profession, but I believe we are very close to making a change, is if pharmacy were to gain provider status in Illinois. I believe this would be a major step in pushing our profession forward.
Tell us a story about how you selected pharmacy as your profession. How do you know you made the right choice?
In High School I worked at a local drug store stocking shelves. One day the pharmacy cashier called out of work and I was asked to jump in and help with the cash register in pharmacy. This was my first introduction to pharmacy and I instantly was enthralled. I saw the Pharmacist counseling patients on their prescriptions and helping customers with over-the-counter options to assist with their healthcare concerns. I left that day forever changed. I couldn’t become a Pharmacy Technician fast enough and I quickly obtained my PTCB certification. It wasn’t long after becoming a Pharmacy Technician that I knew the profession of Pharmacy was for me. During Pharmacy school I changed from a retail pharmacy technician to a hospital pharmacy technician and a new career aspiration formed. For the first time I was exposed to a clinical pharmacist in a hospital and this was the point in time that I knew I had made the right choice. I knew at this moment that Pharmacy was part of who I am and it solidified that I had made the right choice to become a pharmacist.
What makes ICHP great awesome?
I love that ICHP brings brilliant local minds together to have one singular voice. It is vital for everyone to come together and be represented by ICHP as a unified front. The megaphone that ICHP holds in its hands to proclaim, drive, and instill our values as a profession is invaluable.
What initially motivated you to get involved in ICHP?
In a profession that is rapidly changing I felt it was imperative to be a part of a larger collaborative of like minds to stay aligned on where our profession needs to go. I was motivated by ICHP’s involvement in our profession and I could not help but be a part of it.
Is there an individual that you admire or look up to, or a mentor that has influenced your career?
The current Vice President of Pharmacy at UChicago Medicine - Kevin Colgan has inspired me to become a better pharmacist, a better leader, and a better person. Each day Kevin comes to the table with innovative ideas and a drive not matched by anyone else I have ever met.
What advice would you give to student pharmacists?
Do not box yourself in to a set of rules that are made for you. Each day make sure that you are searching and reaching for new heights. Challenge all assumptions. Break those barriers. Do not become a variable in an equation that yields mediocrity. Be curious. Go down tangents as a student. This is your time to explore your knowledge base and find out where your deficiencies lie. Do not ever let anyone tell you that you cannot achieve greatness because YOU, for sure, can accomplish anything YOU put YOUR mind to.
Do you have any special interests or hobbies outside of work?
My passion is traveling and exploring the world. Traveling has for sure been limited during the pandemic, however it has given me some time to explore places locally in Chicagoland and neighboring states that I have never really paid attention to. The first week of September I decided to dip my toes back into traveling and explore the mountains of Alaska.
What is your favorite restaurant or food?
Growing up in Texas I for sure miss some good Tex Mex and BBQ. If anyone has good restaurant suggestions, please by all means, contact me.
Where is your favorite place to vacation?
Rarely the same place twice.
ICHPeople
New job? Get Married? Have a baby? Win an award?
College Connection
University of Illinois at Chicago, College of Pharmacy
Welcome Back
College Connection
by Evan Fetten, P3 Rockford Co-President University of Illinois at Chicago College of Pharmacy
The University of Illinois at Chicago College of Pharmacy student chapter of ICHP has once again continued its tradition of promoting excellence in pharmacy throughout the 2020-2021 academic year. Our E-board showcased its collective creativity and commitment to realizing ICHP’s mission. We held numerous educational, networking, and fundraising events that served as excellent opportunities for our student body to broaden their pharmacy knowledge, sharpen clinical skills, refine interviewing abilities, improve career prospects, and support humanitarian causes. What we have in store for the current academic year, 2021-2022, is no less ambitious; this year, we have several events lined up that will further help pharmacy students improve their career prospects and preserve the pharmacy profession’s integrity and ultimately improve patient care.
Our general body meetings last year, many of which were held in collaboration with other student organizations, consisted of conversations with guest speakers, including practicing pharmacists, pharmacy residents, P4 students, and others. The topics discussed included overviews of a wide range of health systems pharmacy roles, advice on how to combat burnout in residency programs, how to prudently manage student finances, and how to transition to working in an industry setting from a clinical and academic background as a pharmacist. These all served as excellent opportunities for students to learn about various health-systems pharmacy career paths and how to effectively pursue them. In addition to these meetings, our chapter co-hosted a workshop where students received constructive feedback on their curricula vitae. We also debuted a series of interview workshops where residency program directors and residents provided advice on interviewing for residency programs, and P4 and P1 students performed mock interviews in preparation for their residency, internship, and externship interviews before being given valuable feedback. This series proved to be a great success resulting in many students gaining helpful preparatory experience and guidance on interviewing. Complementing these events was a residency panel where residency directors presented on the different residency programs in the Chicagoland area. This served as an opportunity for prospective residency candidates to learn and inquire about these residency programs. To facilitate up-to-date education on therapeutics, our chapter hosted “Journal Club: LETS Talk” and “Morning Rounds: Cases and Therapeutics” sessions where students reviewed, critiqued, and examined journal articles and therapeutic cases from a variety of practice settings. In addition to these events, our chapter held numerous community service activities. We raised awareness and funds for various issues, including lung cancer, HIV/AIDS, climate change, and rainforest destruction. We also held an internship and externship fair to help first-year students network and inquire about different internships and externships, a mindfulness video series, an eating disorders awareness event, the clinical skills competition sessions, and the ICHP student leadership retreat.
Our chapter has a similar series of events lined up for the current academic year. We plan on hosting all of our usual events plus some new ones. Of particular interest is that we will host a series of activities in December that will raise awareness and funding for a specific health topic. We also hope to bring back the hospital tour series for the P1 students, which has always been an excellent opportunity to learn about different hospital pharmacy practice settings. Additionally, we have many novel ideas for fundraisers that we anticipate will elicit ample engagement. With all of these plans, our goal of realizing ICHP’s mission of promoting excellence in pharmacy will manifest.
Midwestern University College of Pharmacy, Downers Grove
A Year of Online Classes Through a Pandemic
College Connection
by Alex Wu, PS-2 Student Chapter Treasurer Midwestern University College of Pharmacy, Downers Grove Campus
At the start of the pandemic, I was at a heightened emotional state where I was worried and nervous. The idea of languishing came about as this emotional state between suffering and thriving.1 That was how I felt, especially with online classes. I had a significant daily workload, but it was not exciting. I was not excited to show up to Zoom classes and to do what felt like busywork. So I have been doing the minimum, and I am not proud of that.
Recognizing that it is normal to be feeling this way was a difficult reflection. It is not atypical for people to be feeling this languish – naming that emotion has allowed me to address it. Over the past year, I shifted my focus from planning ahead to what is in front of me. Every time I looked forward to something, the rug would get pulled out from underneath. While this is an effective strategy for survival, it is not effective for thriving. I created small goals such as catching up with a friend and tidying up my apartment once a week, which brought me joy. I enjoy and get excited having goals to look forward to with in-person classes and interactions slowly making their way back.
Online schooling has made having boundaries between school and personal life incredibly difficult. I am fortunate to have a separate room, which creates a physical boundary from where I work and rest. However, I lacked those boundaries in other ways, especially technological. Online schooling is tricky when your social life is also online. Your friends and professors are communicating with you through the same device. It was challenging to talk to friends or professors without receiving notifications from the other end. Unhealthy patterns came about, such as not reading the professors’ constructive feedback or jumping out in the middle of a conversation with a friend to read what the professor had to say. This threw me into a loop, and reflecting on this has made me realize the importance of explicit boundaries and how I am not great at creating implicit boundaries intuitively, especially on digital screens. Figuring out clear boundaries, such as turning off notifications at a specific time, has helped create a virtual boundary. In the future, this will become easier as social life returns to being more in-person. You can put your phone in your pocket and forget about it because you are with friends.
Upon reflecting on some tasks I did and did not do well this year, I realize there is a defining characteristic. Namely, the things that I did well were things that I made habits. For example, exercise was something that I habitualized. I decided to go to the gym every morning, and it became a habit. Unfortunately, my sleep suffered because it was something that I did not make a habit. I describe willpower as a depletable battery. Every time you decide to do something, you use part of that battery. Throughout the pandemic, it was tiring having to decide every day if I would do this or that. By habitualizing daily tasks that I once thought were tedious, I established a general routine that helped preserve my battery for the unexpected. Although it is important to recognize the power of habits, it is necessary to have spontaneity and not overthink things where every element of every day is planned. A lot of informal interactions, especially social interactions, tend to be spontaneous.
There is a term called MVP – Minimum Viable Product where you build only the minimum features required to test your hypothesis on whether or not your idea has potential.2 I like to apply this term for each day – what are the minimum viable habits that I need in my life? I have been planning my days by scheduling only the important things and keeping the rest of the time for exploration and spontaneity. I want to do things that will bring me joy and recharge my battery to get me moving forward again. Planning is something you play with yourself. You figure out what is appropriate for you in your current phase in life, and you can only do that through experimentation.
References:
- Grant, Adam. “There’s a Name for the Blah You’re Feeling: It’s Called Languishing.” NYTimes.com, The New York Times, 19 April 2021, https://www.nytimes.com/2021/04/19/well/mind/covid-mental-health-languishing.html. Accessed 21 April 2021.
- Aberant, Josh. “The Power of Minimum Viable Products (and the Key to Their Success).” CMSWire.com, CMSWire, 23 January 2018, https://www.cmswire.com/digital-experience/the-power-of-minimum-viable-products-and-the-key-to-their-success/. Accessed 1 June 2021.
Southern Illinois University Edwardsville School of Pharmacy
Coming Back to In-Person in Full Swing
College Connection
by Khushali Sarnot, P2, President-Elect SIUE School of Pharmacy
The SIUE School of Pharmacy is back in person and in full swing after being online for over eighteen months. After spending so long online, two groups of students are experiencing being in person at the school of pharmacy for the first time. The brand new wave of P1 students is excited to start their experience in the professional school and still adjusting to the rigors and challenges of pharmacy school while the P2 students are getting to know each other and the faculty in person for the first time. The P3 students are the guiding light for the other students as they have the most experience of being on campus with a semester and a half under their belt and are still adjusting. Though for the rest of their peers, they are the ones giving advice and leading the charge with different projects in student organizations as some of them have been on hold for eighteen months.
For every student, being back in person has been a challenge, and it has been an obstacle for student organizations. There are set university rules and outside-of-school mandates that we have to follow to make sure everyone is safe. The Student Society of Health-System Pharmacists’ (SSHP) first general meeting was held at the school of pharmacy in August. Usually, in the past, the student chapter had offered pizza to increase attendance. However, this year the executive board decided to provide individually wrapped sandwiches to practice safe food handling. The executive board also had to be creative about tracking attendance since there was a safety concern in using a sheet that everyone would touch. In response, the exec board decided to go with a QR code projected on the presentation slides that the students could scan on their phones, and it would lead them to a website that would track attendance safely and effectively.
Moving forward, SSHP’s effort to plan events has been made with COVID-19 restrictions in mind. The executive board discussed intensively at the chapter leadership retreat on the first day of school about the year for the students and the organization. The student chapter also had a very informative meeting at the Illinois Council of Health-System Pharmacists’ (ICHP) leadership retreat. Students brainstormed different ideas with other Illinois pharmacy students about activities and events that could be planned. The most exciting events planned this year are the residency happy hours that have a wide selection of professionals coming to talk with students about residency and their experience with the match and application process. These events have been set up to be online and more accessible for students, especially for the P4 students out on rotation.
The COVID-19 pandemic has taught us to adapt to the challenges of online classes, and now we will adapt to being back in person and returning to normal. This adversity has only made the SIUE School of Pharmacy students more creative and innovative in their endeavors. A major takeaway from online classes has been implemented into future SSHP events and activities to connect with more students and create more opportunities.
to students on becoming strong candidates for a future residency position.
We will be taking our foot off of the breaks and putting it back on the accelerator in the new academic year. We plan to bring back our professional, social, philanthropy, and fundraising events at full force and develop our RFUMS ICHP chapter and students. The pandemic didn’t put out our light; it simply made our will stronger to burn.
Features
2021 ICHP Annual Meeting Recap
A Virtual Success
by Melissa Dyrdahl, BA; Communications Manager, ICHP
Another Annual Meeting is in the books! We had truly hoped to be meeting face to face in 2021, but with COVID-19 cases rising across Illinois and the more contagious delta variant running rampant, the ICHP executive board made the tough decision to take us virtual. Once the decision was made, ICHP staff did a complete 180-degree pivot from in-person planning to virtual planning - thankfully, we’d had a few virtual ventures under our belts at this point. It was a true team effort (with the bonus support of student intern Matthew Smith from UIC), and we were all quite pleased with what we were able to pull off in just two and a half weeks.
This year’s meeting included up to 17.75 contact hours of CPE credit for pharmacists and 14.0 contact hours for pharmacy technicians! A total of 328 pharmacists, technicians, and students registered for the meeting and joined us for top-notch educational sessions, the installation of ICHP and affiliate officers, a town hall meeting, and our awards ceremony. We even offered our residency showcase program for students, featuring 25 hospitals and health-systems from around the Midwest and a reverse exhibit for our industry representatives (we capped out at 60 reps!)
Our keynote presentation was a definite showstopper! Dr. Ayesha Khan and Dr. Lalita Prasad-Reddy talked about Precepting and Teaching in 2021 – focusing on implicit bias, macroaggressions, and systemic racism in healthcare. This was one of the few programs we were able to record from this year’s meeting – we’re hoping to offer it to you as a home study to fulfill the new implicit bias credit requirement – more details to come!
The 2021 Annual Meeting marked the transition of Jen Arnoldi’s role of president to immediate-past-president and the start of Kathryn Schultz’s reign as ICHP president (check out her inaugural address in the President’s Message column and soon to be available on our YouTube channel!)
Other new ICHP Board members installed included:
- Treasurer - Bernice Man
- Director, Professional Affairs - Megan Corrigan
- Director, Organizational Affairs - Lara Ellinger
- Director, Government Affairs - Peter Couri
- Northern Region Director - Jason Orr
- Central Region Director - Karin Terry
- Southern Region Director - Erika Diericx
- Chair, New Practitioners Network - Justin Moore
The following affiliate officers were also installed:
Metro East Society Officers:
President - Jared Sheley
Northern Illinois Society officers:
- President - Jason Alegro
- President-elect - Lalita Prasad-Reddy
- Immediate Past President - Andrew Merker
- Treasurer - Brian Cryder
- Technician Representative - Glen Gard
Sangamiss Society Officers:
- President - Kayla DuBois
- President-elect - Katelyn Fryman
- Immediate Past President - Ashlie Kallal
West Central Society Officer:
Rock Valley Society Officer:
In addition, Hina Patel was appointed as chair of ICHP’s Residency Leader’s Network, and Matthew Bizewski was appointed as chair of the Ambulatory Care Network.
Another special moment of the meeting was celebrating those who give their all to ICHP! It was wonderful to be able to award and hear from this year’s award winners. Each one of these award recipients is a true leader in our organization!
ICHP Pharmacist of the Year –
ICHP Amy Lodolce Mentorship Award –
ICHP Honorary Membership –
ICHP Technician of the Year –
ICHP NPN Leadership Award –
ICHP Outstanding Volunteer Award –
ICHP President’s Award –
ICHP Past President’s Award –
ICHP Industry Award –
ICHP Shining Stars -
- Peter Couri
- Bridget Dolan
- Julie Downen
- Mark Greg
- Heather Harper
- Chris Herndon
- Alifiya Hyderi
- Randall Knoebel
- Mary Lee
- Katheryn Madden
- Kayla Muench
- Milena Murray
- Jason Orr
- Jennifer Phillips
- Marianne Pop
- Janki Vyas
- Elise Wozniak
We were also able to present the 2021 Best Practice award to Ron Hartman and his team at OSF Healthcare for their winning program, “Software-Aided Compounding: Adopting Technology to Improve Patient Safety and Documentation.” Patty Kohli, Karin Terry, Liz Harthan, Heather Harper, and Jamie Martin were other contributors to the program. Be sure to take advantage of the homestudy on page 18! The winners received a $1,000 check for their department. Krause Construction sponsored this year’s best practice award!
Speaking of sponsors – no meeting would be possible without contributions from our industry representatives! ICHP uses these funds to help keep meeting attendance registration fees low (and look around at other states…we’ve got GREAT prices for members). We want to send a big thank you to our meeting sponsors (you can find links to their websites on our meeting page:
https://ichpnet.org/events/annual_meeting/.
This year’s Annual Meeting sponsors were:
Diamond
Platinum
Gold
- DrFirst
- Nexus Pharmaceuticals, Inc
- United Pharmacy Staffing
Silver
Another program that keeps attendee costs low is our Reverse Exhibit. This event is always wildly successful in huge part to our Pharmacy Directors! These members graciously give us a large chunk of their workday to make a significant impact on what ICHP can offer its members – so a big round of applause to:
- Matthew Allsbrook
- Erick Borkowski
- Megan Corrigan
- Peter Couri
- Mark Deaton
- Andy Donnelly
- Jeanne Durley
- Tara Gorden
- Mike Guithues
- Luke Herbert
- Rick Jacobson
- Rakesh Jain
- Jason Orr
- William Pong
- Elaine Rodriguez
- Sandy Salverson
- Rishita Shah
- Erin Shaughnessy
- Ehsan Tavassoli
- Jeff Thiel
- Georgeta Titean
- Joe Vadakara
- Thomas Yu
Whether in person or virtual – one thing is for sure – an ICHP Annual Meeting is MUCH MORE than just a way to get your CPE – it’s pharmacy professionals from across the state dedicating their time and energy to advance excellence in the profession. Sound exciting? Want to get involved? Consider joining next year’s Annual Meeting planning committee, submitting a presentation proposal, or even participating in our Spring Meeting poster presentations.
ICHP President Kathryn Schultz’s theme for the upcoming year is “Everybody In” – so what are you waiting for? Jump in!
More
Upcoming Events
CE Appetizer
November 3, 2021
Confronting and Mitigating our Implicit Bias to Promote Health Equity
Sally Arif, PharmD, BCPS, BCCP
Accredited for pharmacists and pharmacy technicians
Sangamiss Live Webinar
November 9, 2021
Well that De-escalated Quickly: A Guide to Antibiotic De-escalation and Early Discontinuation of Therapy
Alina Viteri, PharmD
Accredited for pharmacists only
ICHP Spring Meeting
March 11 & 12, 2022
Embassy Suites
East Peoria, IL
NISHP Meeting & Live Webinar
Open to all ichp members!
December 1, 2021 (Save the date!)
Don't Forget About the Flu: Identifying, Treating, and Protecting High Risk Patients
James S. Lewis II, PharmD, FIDSA
Accredited for pharmacists only
Pharmacy Legislative Day
To be Announced
More details to come...
Welcome New Members!
A big welcome to our new members!
ICHP Pharmacy Action Fund Contributors (PAC)
Thanks to everyone who contributed!

Print Entire Issue


 President's Message
President's Message.jpg)
 Crank's Corner
Crank's Corner