Official Newsjournal of the Illinois Council of Health-System Pharmacists

November 2021
Volume 47 Issue 4
President's Message
Directly Speaking
Features
Call for Entries: 2014 Best Practice Award
Clinical Practice and Research Network Meeting
What I Learned During My Time with ICHP
Columns
College Connections
Mini-Health Fair at the Indo American Center
APPE Preparation through SSHP Quarterly Journal Club Discussions
Second Annual Student and Pharmacist Mixer
More
Officers and Board of Directors
ICHP Pharmacy Action Fund (PAC) Contributors
KeePosted Info

Illinois Council of Health-System Pharmacists
4055 North Perryville Road
Loves Park, IL 61111-8653
Phone: (815) 227-9292
Fax: (815) 227-9294
www.ichpnet.org
KeePosted
Official Newsjournal of the Illinois Council of Health-System Pharmacists
EDITOR
Jacob Gettig
ASSISTANT EDITOR
Jennifer Phillips
MANAGING EDITOR
Scott Meyers
ASSISTANT MANAGING EDITOR
Trish Wegner
DESIGN EDITOR
Amanda Wolff
ICHP Staff
EXECUTIVE VICE PRESIDENT
Scott Meyers
VICE PRESIDENT - PROFESSIONAL SERVICES
Trish Wegner
DIRECTOR OF OPERATIONS
Maggie Allen
INFORMATION SYSTEMS MANAGER
Heidi Sunday
CUSTOMER SERVICE AND
PHARMACY TECH TOPICS™ SPECIALIST
Jo Ann Haley
ACCOUNTANT
Jan Mark
COMMUNICATIONS MANAGER
Amanda Wolff
LEGISLATIVE CONSULTANT
Jim Owen
ICHP Mission Statement
Advancing Excellence in the Practice of Pharmacy
ICHP Vision Statement
ICHP dedicates itself to achieving a vision of pharmacy practice where:
- Pharmacists are universally recognized as health care professionals and essential providers of health care services.
- Patients are aware of the training, skills, and abilities of a pharmacist and the fundamental role that pharmacists play in optimizing medication therapy.
- Formally educated, appropriately trained, and PTCB certified pharmacy technicians manage the medication distribution process with appropriate pharmacist oversight.
- Pharmacists improve patient care and medication safety through the development of effective public policies by interacting and collaborating with patients, other health care professionals and their respective professional societies, government agencies, employers and other concerned parties.
- Evidence-based practices are used to achieve safe and effective medication therapies.
- There are an adequate number of qualified pharmacy leaders within the pharmacy profession.
- Pharmacists take primary responsibility for educating pharmacy technicians, pharmacy students, pharmacist peers, other health professionals, and patients about appropriate medication use.
KeePosted Vision
As an integral publication of the Illinois Council of Health-System Pharmacists, the KeePosted newsjournal will reflect its mission and goals. In conjunction with those goals, KeePosted will provide timely information that meets the changing professional and personal needs of Illinois pharmacists and technicians, and maintain high publication standards.
KeePosted is an official publication of, and is copyrighted by, the Illinois Council of Health-System Pharmacists (ICHP). KeePosted is published 10 times a year. ICHP members received KeePosted as a member benefit. All articles published herein represent the opinions of the authors and do not reflect the policy of the ICHP or the authors’ institutions unless specified. Advertising inquiries can be directed to ICHP office at the address listed above. Image disclaimer: The image used in the Pharmacy Tech Topics™ advertisement is the property of © 2014 Thinkstock, a division of Getty Images.
Copyright © 2014, Illinois Council of Health-System Pharmacists. All rights reserved.
Features
Call for Entries: 2014 Best Practice Award

The objective of the Best Practice Award program is to encourage the development of innovative or creative pharmacy practice programs or innovative approaches to existing pharmacy practice challenges in health systems within the state of Illinois. Applicants will be judged on their descriptions of programs and practices employed in their health system based on the following criteria:
Past winners include: 2013Nicole Rabs, Pharm.D., Sarah M. Wieczorkiewicz, Pharm.D., BCPS, Michael Costello, PhD, and Ina Zamfirova, BA “Development of a Urinary-Specific Antibiogram for Gram Negative Isolates: Impact of Patient Risk Factors on Susceptibility” 2012 2011 |
Online entry form: http://www.ichpnet.org/professional_practice/best_practices/ Eligibility Instructions for preparing manuscript The manuscript should be organized as a descriptive report using the following headings:
Format All applicants will be notified of their status within three weeks of the submission deadline. Should your program be chosen as the winner:
Non-winning submissions may also be considered for publication in the ICHP KeePosted, but your permission will be obtained beforehand. |
Clinical Practice and Research Network Meeting
Transition Between Hospital and Nursing Home
What I Learned During My Time with ICHP
by Amanda Peerboom, 2014 PharmD candidate, Chicago State University College of Pharmacy
Columns
 President's Message
President's Message
Doing the Right Thing
by Mike Fotis, ICHP President
 Directly Speaking
Directly Speaking
All You Really Have To Do Is Ask!
by Scott A. Meyers, Executive Vice President
- Checkbook members
- Users
- Micro-engaged
- Engaged
- Leaders
Educational Affairs
ICHP 2014 Spring Meeting Poster Abstracts
- Alendronate and Clostridium difficile infection: An unusual suspect identified by the FDA Adverse Event Reporting System
- The Effectiveness of a Pharmacist-run Patient Aligned Care Team (PACT) Telephone Clinic Managing Chronic Disease States and Therapeutic Monitoring at a Veterans Affairs Medical Center
- Pharmacists’ role in emergency airway responses
- Safety, effectiveness and cost analysis of rivaroxaban versus fondaparinux for thromboprophylaxis after joint replacement at an inpatient rehabilitation facility
- Intravenous batched medication waste management: a retrospective efficiency review
- Improvement of medication delivery through the use of decentralized pharmacy technicians
- Student Capstone Research Experience: A Five Year Perspective
- Prospective order review by Emergency Department (ED) pharmacists: Enhancing patient safety and aligning with regulatory compliance standards
- Evaluation of the accuracy and completeness of nurse driven admission medication histories after modifications to current practice
- Improving the Pharmacist Orientation Program at a Large Medical Center
- Impact of pharmacist led discharge counseling on 30-day readmissions and emergency department visits
- Pharmacist Involvement in the Medication Management of an Acute Care of Elderly (ACE) Unit
- Evaluation of an institution specific cellulitis antimicrobial guideline
- Pharmacist-led development of an interdisciplinary pain management team within a community hospital setting
- Clinical Outcomes in HIV+ Adults with K65R Mutation
- Evaluating the clinical impact of a computerized physician order entry (CPOE) sepsis bundle order set
- Characterization of Drug Shortages by Mining a Drug Information Service Database
- Nuts and bolts of building new service-lines: Blueprints for establishing an outpatient pharmacy
- Secondary Prevention Medication Prescription Filling Following an Acute Ischemic Stroke and the Relationship to Hospital Readmission Rates
- Evaluating Hospitalization Rates of Elderly Patients with Diabetes: An Observational Assessment Targeting Antidiabetic Medication Safety
- Achievement of A1C, Lipid, and Blood Pressure Goals in a Free Community Clinic
- Evaluating hospitalization rates of elderly patients with potential medication-induced geriatric syndromes: An observational assessment targeting safe prescribing practices
- Extended-infusion piperacillin-tazobactam vs. traditional dosing for improving patient outcomes
- Evaluating the impact of a follow-up telephone call program in recently discharged heart failure patients on 30 day readmission rates
- Illinois Prescribers’ Attitude on the Utility of Medical Marijuana
- Comparison of zolpidem to other drugs associated with falls in hospitalized patients
- Chronic Obstructive Pulmonary Disease (COPD) Exacerbation Inpatient Treatment: A Retrospective Chart Review
The GAS From Springfield
A very successful Legislative Day!
by Jim Owen and Scott Meyers
| Bill No. | Sponsor | Summary | Status | Position |
| SB1454 | Delgado, D-Chicago | Amends the Wholesale Drug Distribution Licensing Act. Provides that notwithstanding any other provision of law, a distributor licensed and regulated by the Department of Financial and Professional Regulation, and registered and regulated by the United States Drug Enforcement Administration, shall be exempt from the storage, reporting, ordering, record keeping and physical security control requirements for Schedule II controlled substances with regard to any material, compound, mixture or preparation containing Hydrocodone. These Controlled Substances shall be subject to the same requirements as those imposed for Schedule III controlled substances. Amends the Illinois Controlled Substances Act. Defines Prescription Monitoring Program Advisory Committee and electronic health record. Provides that Dihydrocodeinone (Hydrocodone) with one or more active, non-narcotic ingredients in regional therapeutic amounts is a Schedule III controlled substance, subject to the requirements for prescribing of Schedule III controlled substances with the exception that any prescription must be limited to no more than a 30-day supply with any continuation requiring a new prescription. Provides that prescribers may issue multiple prescriptions (3 sequential 30-day supplies) for Dihydrocodeinone (Hydrocodone), authorizing up to a 90-day supply. Provides that by January 1, 2018, all Electronic Health Records Systems should interface with the Prescription Monitoring Program application program interface to insure that all providers have access to specific patient records as they are treating the patient. Makes other changes. | Passed Senate as Amended; Rules Committee in the House | Neutral |
| SB1934 | Munoz, D-Chicago | Amends the Pharmacy Practice Act. Provides that a pharmacist may substitute a prescription biosimilar product for a prescribed biological product under certain circumstances. Provides that the Board shall adopt rules for compliance with these provisions. | Assignments | Oppose |
| SB2585 | Kotowski, D- Park Ridge | Amends the Illinois Public Aid Code and the Illinois Insurance Code. Requires the Department of Healthcare and Family Services and the Department of Insurance to jointly develop a uniform prior authorization form for prescription drug benefits on or before July 1, 2014. Provides that on and after January 1, 2015, or 6 months after the form is developed, whichever is later, every prescribing provider may use that uniform prior authorization form to request prior authorization for coverage of prescription drug benefits and every health care service plan shall accept that form as sufficient to request prior authorization for prescription drug benefits. Provides that on and after January 1, 2015, a health insurer that provides prescription drug benefits shall utilize and accept the prior authorization form when requiring prior authorization for prescription drug benefits; and that if a health care service plan fails to utilize or accept the prior authorization form, or fails to respond within 2 business days upon receipt of a completed prior authorization request from a prescribing provider, the prior authorization request shall be deemed to have been granted. Exempts certain providers. Sets forth certain criteria for the prior authorization form. Provides that "prescribing provider" includes a provider authorized to write a prescription as described in the Pharmacy Practice Act. Effective January 1, 2014. | Human Services Committee | Oppose |
| SB2674 | Harmon, D- Oak Park | Amends the State Finance Act. Adds the Poison Response Fund. Amends the Wireless Emergency Telephone Safety Act. Provides that human poison control centers constitute an enhancement to 9-1-1 services pursuant to federal law. Provides that for surcharges collected and remitted on or after July 1, 2013, $0.1275 per surcharge collected shall be deposited into the Wireless Carrier Reimbursement Fund on the last day of each month, $0.5825 per surcharge collected shall be deposited into the Wireless Service Emergency Fund, $0.02 per surcharge shall be deposited in the Poison Response Fund, and $0.01 per surcharge collected may be disbursed to the Illinois Commerce Commission for administrative costs. Requires the Auditor General to conduct an annual audit of the Poison Response Fund. Permits the Commission to require an annual report of income and expenditures from each human poison control center. Extends the date of repeal of the Act to July 1, 2018 (currently July 1, 2013). Creates the Poison Response Fund. Amends the Public Utilities Act. Extends the repeal of certain Sections relating to 9-1-1 system providers until July 1, 2016 (currently July 1, 2015). Effective immediately. | 2nd Reading in the Senate | Support |
| SB2941 | Raoul, D-Chicago | Amends the Criminal Identification Act. Authorizes the court to seal Class 4 felony convictions for possession with intent to manufacture or deliver cannabis without the defendant being required to successfully complete qualified probation under the Act. Authorizes the court to seal Class 3 felony convictions for possession with intent to manufacture or deliver cannabis without the defendant being required to obtain an authorization for sealing from the Prisoner Review Board. This would blind potential employers who are required to do a background check on potential hires. Effective immediately. | Passed the Senate; Rules Committee in the House | Oppose |
| SB3109 | McGuire, D-Crest Hill | Amends the Illinois Optometric Practice Act of 1987. Permits a licensed optometrist to prescribe Dihydrocodeinone (Hydrocodone) with one or more active, non-narcotic ingredients only in a quantity sufficient to provide treatment for up to 72 hours, and only if such formulations are reclassified as Schedule II by the U.S. Food and Drug Administration. | 2nd Reading in the Senate | Oppose |
| SB3277 | Althoff, R-McHenry | Amends the Pharmacy Practice Act. Adds the administration of the Meningococcal vaccine to patients 10 through 13 years of age to the definition of "practice of pharmacy". | Licensed Activities Committee | Support |
| SB3502 | Koehler, D- Peoria | Amends the Illinois Controlled Substances Act. Provides that substances containing ephedrine or pseudoephedrine, their salts or optical isomers, or salts of optical isomers, are Schedule III controlled substances and require a prescription. | Criminal Law Committee | Oppose |
| HB3638 | Fine, D- Glenview | Amends the Illinois Public Aid Code and the Illinois Insurance Code. Requires the Department of Healthcare and Family Services and the Department of Insurance to jointly develop a uniform prior authorization form for prescription drug benefits on or before July 1, 2014. Provides that on and after January 1, 2015, or 6 months after the form is developed, whichever is later, every prescribing provider may use that uniform prior authorization form to request prior authorization for coverage of prescription drug benefits and every health care service plan shall accept that form as sufficient to request prior authorization for prescription drug benefits. Provides that on and after January 1, 2015, a health insurer that provides prescription drug benefits shall utilize and accept the prior authorization form when requiring prior authorization for prescription drug benefits; and that if a health care service plan fails to utilize or accept the prior authorization form, or fails to respond within 2 business days upon receipt of a completed prior authorization request from a prescribing provider, the prior authorization request shall be deemed to have been granted. Exempts certain providers. Sets forth certain criteria for the prior authorization form. Provides that "prescribing provider" includes a provider authorized to write a prescription as described in the Pharmacy Practice Act. Effective January 1, 2014. | 2nd Reading in the House | Oppose |
| HB4230 | Lilly, D-Chicago | Amends the State Finance Act. Adds the Poison Response Fund. Amends the Wireless Emergency Telephone Safety Act. Provides that human poison control centers constitute an enhancement to 9-1-1 services pursuant to federal law. Provides that for surcharges collected and remitted on or after July 1, 2013, $0.1275 per surcharge collected shall be deposited into the Wireless Carrier Reimbursement Fund on the last day of each month, $0.5825 per surcharge collected shall be deposited into the Wireless Service Emergency Fund, $0.02 per surcharge shall be deposited in the Poison Response Fund, and $0.01 per surcharge collected may be disbursed to the Illinois Commerce Commission for administrative costs. Requires the Auditor General to conduct an annual audit of the Poison Response Fund. Permits the Commission to require an annual report of income and expenditures from each human poison control center. Extends the date of repeal of the Act to July 1, 2018 (currently July 1, 2013). Creates the Poison Response Fund. Amends the Public Utilities Act. Extends the repeal of certain Sections relating to 9-1-1 system providers until July 1, 2016 (currently July 1, 2015). Effective immediately. | 2nd Reading in the House | Support |
| HB4484 | Reboletti, R-Addison | Creates the Patient Transitions and Continuity of Care Act. Provides that whenever a patient is transferred from a hospital, nursing home, or assisted living facility the transferring hospital, nursing home, or assisted living facility shall provide the receiving hospital, nursing home, or assisted living facility with a form that lists certain specified information about the patient. Provides that the Illinois Department of Public Health shall develop and publish the form that is to be used by the transferring hospital, nursing home, or assisted living facility. Effective immediately. | Rules Committee | Support |
| HB4575 | Lilly, D-Chicago | Appropriates $1,331,100 from the General Revenue Fund to the Department of Public Health from the General Revenue Fund for grants to the Illinois Poison Center. Effective July 1, 2014. | Rules Committee | Support |
| HB4580 | Lilly, D-Chicago | Amends the Health Care Worker Background Check Act. Provides that the prohibition against a health care employer or long-term care facility hiring, employing, or retaining an individual in a position with duties involving direct care for clients, patients, or residents, or duties that involve or may involve contact with long-term care facility residents or access to the living quarters or the financial, medical, or personal records of residents, on account of the individual's conviction of committing or attempting to commit one or more of certain specified offenses shall be for a period of (i) 2 years following the date of conviction in the case of a conviction for a misdemeanor and (ii) 5 years following the date of conviction in the case of a conviction for a felony. Effective immediately. | Rules Committee | Neutral |
| HB5631 | Gabel, D-Evanston | Amends the Pharmacy Practice Act. Defines "bleeding disorder", "blood clotting product", and "established patient". Establishes certain requirements, standards of care, and business practices that pharmacies and pharmacists shall comply with when dispensing blood clotting products. | Health Care Licenses Committee | Oppose |
| HB5924 | Zalewski, D-Riverside | Amends the Illinois Vehicle Code. Provides that a person shall not drive or be in actual physical control of any vehicle within this State while there is any amount of a drug, substance, or compound in the person's breath, blood, or urine resulting from the use or consumption of a controlled substance listed in the Illinois Controlled Substances Act in excess of the prescribed amount in the person's prescription for the controlled substance. | Judiciary Committee | Oppose |
| HB5987 | Phelps, D-Harrisburg | Creates the Audits of Pharmacy Benefits Act. Imposes a number of requirements on audits of pharmacy services conducted pursuant to a contract entered into by the pharmacy and the auditing entity on behalf of a health carrier or a pharmacy benefits manager. Requires the entity conducting a pharmacy audit to deliver a preliminary audit report to the pharmacy and to give the pharmacy an opportunity to respond to the report prior to issuing a final audit report. Provides that the entity is also required to implement a process for appealing the findings of the final audit report, and further provides that if either party is unsatisfied with the appeal, that party may seek relief under the terms of the contract. Establishes a number of requirements that the auditing entity must follow when calculating the amounts and penalties that are to be recovered from the pharmacy based on the audit report, and prohibits the entity from receiving payment on any basis tied to the amount claimed or recovered from the pharmacy. Effective immediately. | Rules Committee | Oppose in current form |
ICHPeople
 Congratulations to Dr. Jennifer D'Souza on being chosen to participate in ASHP's Research and Education Foundation Research Boot Camp! Below is the official press release.
Congratulations to Dr. Jennifer D'Souza on being chosen to participate in ASHP's Research and Education Foundation Research Boot Camp! Below is the official press release. Congratulations to Mary Ann Kliethermes whose book, Building a Successful Ambulatory Care Practice: A Complete Guide for Pharmacists, is published and available in the ASHP Store!
Congratulations to Mary Ann Kliethermes whose book, Building a Successful Ambulatory Care Practice: A Complete Guide for Pharmacists, is published and available in the ASHP Store! Congratulations to Megan Metzke who welcomed son, Drake Andrew Metzke, with her husband, Brian, on December 20, 2013.
Congratulations to Megan Metzke who welcomed son, Drake Andrew Metzke, with her husband, Brian, on December 20, 2013. Congratulations to Lisa (Lubsch) Bimpasis who welcomed son, Alexander Michael Bimpasis, with her husband, Nick, on February 13, 2014.
Congratulations to Lisa (Lubsch) Bimpasis who welcomed son, Alexander Michael Bimpasis, with her husband, Nick, on February 13, 2014.
Leadership Profile
David Tjhio
 Where did you go to pharmacy school?
Where did you go to pharmacy school?
Medication Safety Pearl
Clinical Informatics and Medication Safety: Partnering for a Safer EMR
by Heather Harper, PharmD, BCPS and Karin Terry, PharmD, Clinical Informatics Pharmacist and Micro-Medication Safety Officer, OSF Saint Francis Medical Center, Peoria, IL
- Campbell EM, Sittig DF, Ash JS, Guappone KP, Dykstra RH. Types of unintended consequences related to computerized provider order entry. J Am Med Inform Assoc. 2006; 13(5): 547-556.
- Koppel R, Metlay JP, et al. Role of computerized physician order entry systems in facilitating medication errors (abstract). JAMA. 2005; 293(10): 1197–1203.
Board of Pharmacy Update
Highlights of the March Meeting
by Scott A. Meyers, Executive Vice President
- Pharmacists may be involved in the dispensing process of medical cannabis.
- Pharmacies may not apply for or be medical cannabis dispensaries in Illinois.
- Many sections on the application for a dispensary permit favor the knowledge and skills of pharmacists, relating to medical knowledge, product security, patient counseling and more.
- The Department of Public Health will convene a Medical Cannabis Review Board, and one of the positions on the board is specifically identified as a pharmacist.
New Practitioners Network
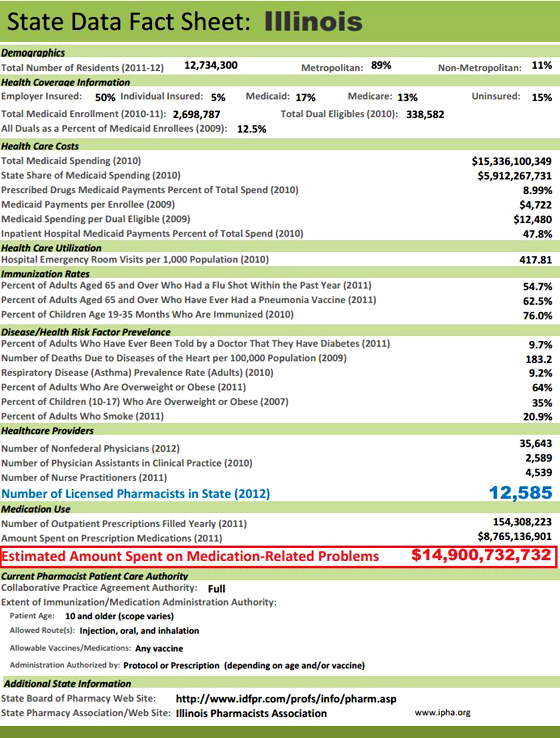
What Does Provider Status Truly Mean?
by Kunal Desai, PharmD Candidate 2014, Chicago State University College of Pharmacy; reviewed by Rebecca Castner, PharmD, Assistant Professor of Pharmacy Practice at Chicago State University COP/Clinical Pharmacy Specialist, Ambulatory Care at Rush University Medical Center
- American Pharmacists Association. The pursuit of provider status: what pharmacists need to know. APhA provider status Q&A informational sheet. http://www.pharmacist.com/sites/default/files/files/Provider%20Status%20FactSheet_Final.pdf (Accessed 2014 Jan 16).
- Academy of Managed Care Pharmacy. Non-physician provider status for pharmacists. Where we stand on non-physician provider status for pharmacists. http://www.amcp.org/Sec.aspx?id=15415 (Accessed 2014 Jan 16).
- National Alliance of State Pharmacy Associations. Patient access to pharmacists’ patient care services: the key to improving medication use and lowering health care costs. 2013 state fact sheets. http://www.naspa.us/documents/facts/2013%20State%20Sheet%20Illinois.pdf (Accessed 2014 Jan 17).
- Centers for Medicare & Medicaid Services. Accountable care organizations (ACO). Baltimore, MD: Centers for Medicare & Medicaid Services, 2013. http://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/ACO/index.html?redirect=/ACO/ (Accessed 2014 Jan 17).
- Yap, D. California provider status law effective January 1. American Pharmacists Association. http://www.pharmacist.com/california-provider-status-law-effective-january-1 (Accessed 2014 Jan 17). News.
- Doctors and Pharmacists Battle Over Scope of Practice. Medscape. Apr 05, 2012.
- Watkins JL, Landgraf A, Barnett CM, Michaud L. Evaluation of pharmacist-provided medication therapy management services in an oncology ambulatory setting. J Am Pharm Assoc (2003). 2012;52(2):170-4.
College Connections
Mini-Health Fair at the Indo American Center
by Zachary Stewart, P3, ICHP Philanthropy Chair, Midwestern University Chicago College of Pharmacy
 The ICHP student chapter at Midwestern University Chicago College of Pharmacy had the opportunity to hold a mini-health fair at the Indo-American Center, a community center based in Chicago, Illinois. This event focused on the importance of medication adherence and diabetes education. The regular center attendees face various challenges in the healthcare system, some of which include language barriers (their main speaking language is Hindi) and lower health literacy. We believed that this population would greatly benefit from a mini-health fair that incorporated multiple healthcare disciplines to help them overcome these obstacles.
The ICHP student chapter at Midwestern University Chicago College of Pharmacy had the opportunity to hold a mini-health fair at the Indo-American Center, a community center based in Chicago, Illinois. This event focused on the importance of medication adherence and diabetes education. The regular center attendees face various challenges in the healthcare system, some of which include language barriers (their main speaking language is Hindi) and lower health literacy. We believed that this population would greatly benefit from a mini-health fair that incorporated multiple healthcare disciplines to help them overcome these obstacles.
Volunteering with Care
by Joanna Kasper, P-3, SSHP Philanthropy Chair, Rosalind Franklin University of Medicine and Science College of Pharmacy
APPE Preparation through SSHP Quarterly Journal Club Discussions
by Kim Rusche P3, SSHP Journal Club Chair, Roosevelt University College of Pharmacy
Second Annual Student and Pharmacist Mixer
by Patrick Costello, PS3, University of Illinois at Chicago College of Pharmacy, Rockford Campus
 As the second class to have the pleasure of attending the UIC Rockford College of Pharmacy, we have benefited greatly from the class ahead of us. Much like an older sibling, they have given us pearls of wisdom, been the guinea pigs, and set up many organizations and events. One such event was the ICHP Student and Pharmacist Mixer. In the fall of 2012, the first ever mixer took place without a hitch, with many area clinical pharmacists in attendance. This was a unique opportunity for our students to both network and learn more about the profession of pharmacy.
As the second class to have the pleasure of attending the UIC Rockford College of Pharmacy, we have benefited greatly from the class ahead of us. Much like an older sibling, they have given us pearls of wisdom, been the guinea pigs, and set up many organizations and events. One such event was the ICHP Student and Pharmacist Mixer. In the fall of 2012, the first ever mixer took place without a hitch, with many area clinical pharmacists in attendance. This was a unique opportunity for our students to both network and learn more about the profession of pharmacy. More
Officers and Board of Directors
Immediate Past President
224-948-1528
tom_westerkamp@baxter.com
President-Elect
linda.fred@carle.com
GINGER ERTEL
Treasurer
660-342-5022
gertel@msn.com
CAROL HEUNISCH
Secretary
847-933-6811
cheunisch@northshore.org
TRAVIS HUNERDOSSE
Director, Educational Affairs
thunerdo@nmh.org
JENNIFER ELLISON
Director, Marketing Affairs
Jennifer.C.Ellison@osfhealthcare.org
DESI KOTIS
Director, Professional Affairs
Director, Government Affairs
MIKE WEAVER
Chairman, House of Delegates
815-599-6113
mweaver@fhn.org
ELIZABETH ENGEBRETSON
Technician Representative
815-756-1521x153346
EEngebretson@northshore.org
DAVID TJHIO
Chairman, Committee on Technology
816-885-4649
david.tjhio@cerner.com
DIANA ISAACS
Chairman, New Practitioners Network
disaacs@csu.edu
JACOB GETTIG
Editor & Chairman, KeePosted Committee
630-515-7324 fax: 630-515-6958
jgetti@midwestern.edu
Assistant Editor, KeePosted
630-515-7167
jphillips@midwestern.edu
KATHRYN SCHULTZ
Regional Director North
kathryn_schultz@rush.edu
JENNIFER ARNOLDI
Regional Director Central
jennifer.arnoldi@st-johns.org
EMMA CARROLL
President, Student Chapter
University of IL C.O.P.
emmacarroll620@gmail.com
Student Chapter Liaison
University of IL C.O.P.
bucek@uic.edu
NADIYAH CHAUDHARY
President, Rockford Student Chapter
University of IL C.O.P.
nadi925@gmail.com
JANEY YU
President, Student Chapter
janey.yu@mwumail.midwestern.edu
BERNICE MAN
President, Student Chapter
Chicago State University C.O.P.
bman@csu.edu
TRAMAINE HARDIMON
Student Representative
Chicago State University C.O.P
thardimo@csu.edu
ZAK VINSON
President, Student Chapter
Southern Illinois University Edwardsville S.O.P
zvinson@siue.edu
ALEX MERSCH
President, Student Chapter
Roosevelt University C.O.P.
amersch@mail.roosevelt.edu
JENNIFER AGUADO
President, Student Chapter
Rosalind Franklin University C.O.P.
jennifer.aguado@my.rfums.org
SCOTT MEYERS
Executive Vice President, ICHP Office
815-227-9292
scottm@ichpnet.org
ICHP AFFILIATES
PETE ANTONOPOULOS
President, Northern IL Society (NISHP)
JULIA SCHIMMELPFENNIG
President, Metro East Society (MESHP)
jschimmelpfen@sebh.org
MEGAN METZKE
President, Sangamiss Society
memiller8@yahoo.com
ED RAINVILLE
President, West Central Society (WSHP)
309-655-7331x
ed.c.rainville@osfhealthcare.org
Vacant Roles at Affiliates —
President, Rock Valley Society; Southern IL Society; Sugar Creek Society; Regional Director, South
Welcome New Members!

| New Member | Recruiter | ||
| Jeffry Ellis | John Maxwell | ||
| Jessica Nacar | |||
| Brittany Huff | |||
| Kerri Eckerling | Mike Fotis | ||
| D'Yana Conley | |||
| Crystal Ellison | |||
| Christine Jeng | |||
| Ozioma Nwaobia | |||
| Fadumo Mire | |||
| Yesol Kim | |||
| Coty Tunwar | |||
| Lila Ahmed | |||
| Marcella Wheatley | Ina Henderson | ||
| L. Amulya Murthy | |||
| Michaela Smith | |||
| Lina Kishta | |||
| Olga Volozhina | |||
| Kimberly Scott | |||
| Magdalena Gacek | |||
| Samantha Plencner | |||
| Linh Do | |||
| Jontia Gentry | |||
| Sung Seo | |||
| Hanifath Lawani | |||
| Frederique Coulibaly | |||
| Sodiq Ogunnaike | |||
| ChungYun Kim | |||
| Allie Habhab | |||
| Michael Lewis | |||
| Gunny Chadha | |||
| Amanda Firmansyah | |||
| Hanh Nguyen | |||
| Greta Musaraj | |||
| Jennifer Ratliff | |||
| Joseph Gomes | |||
|
Carrie Sincak |
ICHP Pharmacy Action Fund (PAC) Contributors
Edward Donnelly
Scott Bergman
Kevin Colgan
Ginger Ertel
Michael Rajski
Michael Weaver
Thomas Westerkamp
Rauf Dalal
GENERAL ASSEMBLY GUILD - $250-$499
Margaret Allen
Janette Mark
Edward Rainville
Heidi Sunday
SPRINGFIELD SOCIETY - $100-$249
Jennifer Arnoldi
Susan Berg
CONTRIBUTOR - $1-$49
Upcoming Events

Chicago, IL
Adventist LaGrange Memorial Hospital | LaGrange, IL
Live Program: University of Illinois at Chicago College of Pharmacy | Chicago, IL
Wrigley Field | Chicago, IL
LIVE Webinar
Oakbrook Terrace, IL
Location TBD

